Evolutionary transcriptomics implicates HAND2 in the origins of implantation and regulation of gestation length
Abstract
The developmental origins and evolutionary histories of cell types, tissues, and organs contribute to the ways in which their dysfunction produces disease. In mammals, the nature, development and evolution of maternal-fetal interactions likely influence diseases of pregnancy. Here we show genes that evolved expression at the maternal-fetal interface in Eutherian mammals play essential roles in the evolution of pregnancy and are associated with immunological disorders and preterm birth. Among these genes is HAND2, a transcription factor that suppresses estrogen signaling, a Eutherian innovation allowing blastocyst implantation. We found dynamic HAND2 expression in the decidua throughout the menstrual cycle and pregnancy, gradually decreasing to a low at term. HAND2 regulates a distinct set of genes in endometrial stromal fibroblasts including IL15, a cytokine also exhibiting dynamic expression throughout the menstrual cycle and gestation, promoting migration of natural killer cells and extravillous cytotrophoblasts. We demonstrate that HAND2 promoter loops to an enhancer containing SNPs implicated in birth weight and gestation length regulation. Collectively, these data connect HAND2 expression at the maternal-fetal interface with evolution of implantation and gestational regulation, and preterm birth.
Introduction
The ontogeny and evolutionary history of cell types, tissues, and organ systems, as well as the life histories of organisms bias the ways in which dysfunctions in those systems underlie disease (Varki, 2012). Thus, a mechanistic understanding of how cells, tissues, and organs evolved their functions, and how organism’s life histories influence them, may provide clues to the molecular etiologies of disease. The most common way of utilizing evolutionary information to characterize the genetic architecture of disease is to link genetic variation within a species to phenotypes using quantitative trait loci (QTL) or genome-wide association studies (GWAS). An alternative approach is to identify fixed genetic differences between species that are phylogenetically correlated with different disease relevant phenotypes. While the risk of cancer increases with the age of an individual, for example, the prevalence of cancer types varies by species (Abegglen et al., 2015), likely because of differences in genetic susceptibility to specific cancers, structure of organ and tissue systems, and life exposures to carcinogens (Varki and Varki, 2015). Similarly, the risk of cardiovascular disease (CVD) increases with age across species, but the pathophysiology of CVD can differ even between closely related taxa such as humans, in which CVD predominantly results from coronary artery atherosclerosis, and the other Great Apes (Hominids), in which CVD is most often associated with interstitial myocardial fibrosis (Varki et al., 2009).
Extant mammals span major stages in the origins and diversification of pregnancy, thus a mechanistic understanding of how pregnancy originated and diverged may provide unique insights into the ontogenetic origins of pregnancy disorders. The platypus and echidna (Monotremes) are oviparous, but the embryo is retained in the uterus for 10–22 days, during which the developing fetus is nourished by maternal secretions delivered through a simple placenta, prior to the laying of a thin, poorly mineralized egg that hatches in ~2 weeks (Hill, 1936). Live birth (viviparity) evolved in the stem-lineage of Therian mammals, but Marsupials and Eutherian (‘Placental’) mammals have dramatically different reproductive strategies. In Marsupials, pregnancies are generally short (~25 days) and completed within the span of a single estrous cycle (Renfree and Shaw, 2001; Renfree, 2010). Eutherians, in contrast, evolved a suite of traits that support prolonged pregnancies (up to 670 days in African elephant), including an interrupted estrous cycle, which allows for gestation lengths longer than a single reproductive cycle, maternal-fetal communication, maternal recognition of pregnancy, implantation of the blastocyst and placenta into uterine tissue, differentiation (decidualization) of endometrial stromal fibroblasts (ESFs) in the uterine lining into decidual stromal cells (DSCs), and maternal immunotolerance of the antigenically distinct fetus, that is the fetal allograft (Guleria and Pollard, 2000; Moffett and Loke, 2004; Erlebacher, 2013).
Gene expression changes at the maternal-fetal interface underlie evolutionary differences in pregnancy (Hou et al., 2012; Lynch et al., 2015; Armstrong et al., 2017), and thus likely also pathologies of pregnancy such as infertility, recurrent spontaneous abortion (Kosova et al., 2015), preeclampsia (Elliot, 2017; Arthur, 2018; Varas Enriquez et al., 2018), and preterm birth (Plunkett et al., 2011; Swaggart et al., 2015; LaBella, 2019). Here, we assembled a collection of gene expression data from the pregnant/gravid maternal-fetal interface of tetrapods and used evolutionary methods to reconstruct gene expression changes during the origins of mammalian pregnancy. We found that genes that evolved to be expressed at the maternal-fetal interface in the Eutherian stem-lineage were enriched for immune functions and diseases, as well as preterm birth. Among the recruited genes was the transcription factor Heart- and neural crest derivatives-expressed protein 2 (HAND2), which plays essential roles in neural crest development (Srivastava et al., 1997), cardiac morphogenesis (Srivastava et al., 1997; Shen et al., 2010; Tamura et al., 2013; Lu et al., 2016; Sun et al., 2016), and suppressing estrogen signaling during the period of uterine receptivity to implantation (Huyen and Bany, 2011; Li et al., 2011; Shindoh et al., 2014; Fukuda et al., 2015; Mestre-Citrinovitz et al., 2015; Murata et al., 2019; Šućurović et al., 2020). We determined that HAND2 expression at the first trimester maternal-fetal interface was almost entirely restricted to cell types in ESF lineage and is regulated by multiple transcription factors that control progesterone responsiveness. Moreover, the HAND2 promoter loops to an enhancer with single-nucleotide polymorphisms (SNPs) that have been implicated by GWAS in the regulation of gestation length (Warrington et al., 2019; Sakabe et al., 2020). Furthermore, we showed that HAND2 regulates interleukin 15 (IL15) expression in ESFs, and that ESF-derived IL15 influences the migration of natural killer and trophoblast cells. These data suggest that HAND2 and IL15 signaling played an important role in the evolution of implantation and regulation of gestation length.
Results
Genes that evolved endometrial expression in Eutherian mammals are enriched in immune functions
We previously used comparative transcriptomics to reconstruct the evolution of gene expression at the maternal-fetal interface during the origins of mammalian pregnancy (Lynch et al., 2015). Here, we assembled a collection of new and existing transcriptomes from the pregnant/gravid endometria of 15 Eutherian mammals, 3 Marsupials, 1 Monotreme (platypus), 2 birds, 5 lizards, and 1 amphibian (Figure 1A and Figure 1—source data 1). The complete dataset includes expression information for 21,750 genes from 27 species. Next, we transformed continuous transcript abundance estimates values into discrete character states such that genes with Transcripts Per Million (TPM) ≥ 2.0 were coded as expressed (state = 1), genes with TPM < 2.0 were coded as not expressed (state = 0), and genes without data in specific species were coded as missing (state = ?). We then used parsimony to reconstruct ancestral transcriptomes and trace the evolution of gene expression gains (0 → 1) and losses (1 → 0) in the endometrium (Figure 1—source data 2).
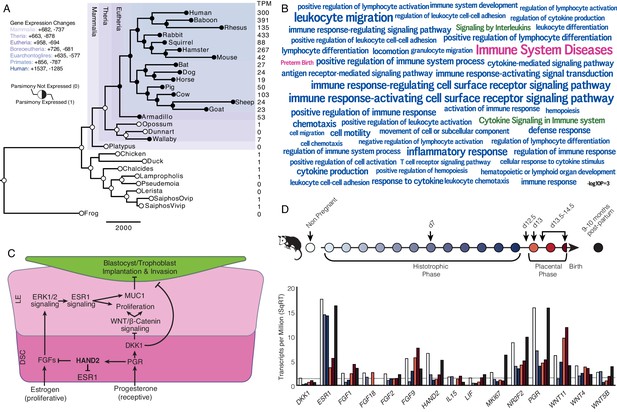
Recruitment of HAND2-mediated anti-estrogenic signaling in the Eutherian endometrium.
(A) Evolution of HAND2 expression at the maternal-fetal interface. Amniotes phylogeny with horizontal branch lengths drawn proportional to the number of gene expression changes inferred by parsimony (most parsimonious reconstruction). Circles indicate HAND2 expression in extant species and ancestral reconstructions. Black, expressed (state = 1). White, not expressed (state = 0). Inset legend shows the number of most gene expression changes from the root node to human (+ = gene expression gained; - = gene expression lost). Numbers to the right indicate HAND2 expression in TPM for each respective species. (B) WordCloud of biological pathways (green), human disease phenotypes (pink), and biological process gene ontology terms (blue) enriched among 149 unambiguously recruited genes in the Eutherian stem-lineage. Term size is shown scaled to -log10 p-value (see inset scale). (C) Cartoon model of estrogen signaling and HAND2-mediated anti-estrogenic signaling in the endometrium. The estrogen-mediated signaling network is suppressed by progesterone through the activation of HAND2 and antagonists of canonical WNT/β-catenin-mediated signaling pathways such as DKK1. In the proliferative phase of the reproductive cycle, estrogen acts through ESR1 in stromal cells to increase the production of fibroblast growth factors (FGFs), which serve as paracrine signals leading to sustained proliferation of epithelial cells. Active estrogen signaling maintains epithelial expression of Mucin 1 (MUC1), a cell surface glycoprotein that acts as a barrier to implantation. During the receptive phase of the cycle, however, progesterone induces HAND2 and DKK1 expression in the endometrial stroma, inhibiting production of FGFs, suppressing epithelial proliferation and antagonizing estrogen-mediated expression of MUC1, thereby promoting uterine receptivity to implantation. DSC = decidual stromal cells, LE = luminal epithelium. (D) Gene expression time course through opossum pregnancy. Upper, schematic of gestation length in Monodelphis domestica in which the histotrophic phase lasts from day 1 to day 12, hatching occurs on day 12.5, the placental phase lasts from day 13 to day 14.5, and birth occurs on day 14.5. Lower, data shown as square root (SqRT) transformed TPM. The TPM = 2 expression cutoff is shown as a horizontal gray line. M. domestica RNA-Seq data from Lynch et al., 2015; Hansen et al., 2016; Griffith et al., 2017; Griffith et al., 2019.
-
Figure 1—source data 1
Species and gene expression information.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data1-v2.xlsx
-
Figure 1—source data 2
Binary encoded endometrial gene expression dataset.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data2-v2.nex.txt
-
Figure 1—source data 3
Genes (HUGO gene names) that unambiguously evolved endometrial expression in the Eutherian stem-lineage (‘Recruited Genes’).
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data3-v2.xlsx
-
Figure 1—source data 4
Top 100 pathways (Wikipathway, Reactome, KEGG) in which Eutherian recruited genes are enriched.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data4-v2.xlsx
-
Figure 1—source data 5
Top 100 human phenotype (disease) ontology terms in which Eutherian recruited genes are enriched.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data5-v2.xlsx
-
Figure 1—source data 6
Top 100 biological process gene ontology (GO) terms in which Eutherian recruited genes are enriched.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data6-v2.xlsx
-
Figure 1—source data 7
RNA-Seq data from opossum endometrial samples.
Gene expression in TPM. NP = non-pregnant; d7, d12.5, d13, d13.5–14.5 = day 7, 12.5, 13, 13.5–14.5 of pregnancy, respectively; N2_9mo, N3_10mo = non-pregnant endometrium 9 and 10 months after pregnancy, respectively.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data7-v2.xlsx
-
Figure 1—source data 8
Database of genes implicated in preterm birth.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig1-data8-v2.gmt.txt
We identified 958 genes that most parsimoniously evolved endometrial expression in the Eutherian stem-lineage, including 149 that unambiguously evolved endometrial expression (Figure 1A and Figure 1—source data 3). These 149 genes were significantly enriched in pathways related to the immune system (Figure 1B and Figure 1—source data 4–6), although only two pathways were enriched at False Discovery Rate (FDR) ≤ 0.10, namely, ‘Cytokine Signaling in Immune System’ (hypergeometric p = 1.97×10−5, FDR = 0.054) and ‘Signaling by Interleukins’ (hypergeometric p = 4.09×10−5, FDR = 0.067). Unambiguously recruited genes were also enriched in numerous human phenotype ontology terms but only two, ‘Immune System Diseases’ (hypergeometric p = 3.15×10−8, FDR = 2.52×10−4) and ‘Preterm Birth’ (hypergeometric p = 4.04×10−4, FDR = 8.07×10−4), were enriched at FDR ≤ 0.10. In contrast, these genes were enriched in numerous biological process gene ontology (GO) terms at FDR ≤ 0.10, nearly all of which were related to regulation of immune system, including ‘Leukocyte Migration’ (hypergeometric p = 1.17×10−7, FDR = 1.29×10−3), ‘Inflammatory Response’ (hypergeometric p = 8.07×10−7, FDR = 2.21×10−3), and ‘Cytokine-mediated Signaling Pathway’ (hypergeometric p = 2.18×10−5, FDR = 0.013).
Recruitment of HAND2 and anti-estrogenic signaling in Eutherians
Among the genes that unambiguously evolved endometrial expression in the Eutherian stem-lineage was the basic helix-loop-helix family transcription factor Heart- and neural crest derivatives-expressed protein 2 (HAND2). HAND2 plays an essential role in mediating the anti-estrogenic action of progesterone and the establishment of uterine receptivity to implantation (Figure 1C; Huyen and Bany, 2011; Li et al., 2011; Fukuda et al., 2015; Mestre-Citrinovitz et al., 2015), suggesting that the silencing of estrogen signaling during the window of implantation is a derived trait in Eutherian mammals. Notably, this silencing is not observed in the pregnant endometrium of Marsupials such as the brush-tailed possum (Trichosurus vulpecula) (Young and McDonald, 1982; Curlewis and Stone, 1987) and tammar wallaby (Macropus eugenii) (Renfree and Blanden, 2000).
To investigate further, we used the short-tailed opossum (Monodelphis domestica) as a model of pregnancy in Marsupials. M. domestica lacks implantation and thus is a good representative of pregnancy in the Therian common ancestor, in contrast to other Marsupials, such as the tammar wallaby, which have derived traits such as delayed ovulation, independently evolved maternal recognition of pregnancy and expression of HAND2 at low levels during gravidity (Figure 1A and Figure 1—figure supplement 1—source data 1, Figure 1—figure supplement 1). Using existing RNA-Seq data from short-tailed opossum endometria, we analyzed a time course consisting of day 7 (during the histotrophic phase), day 12.5 (just after hatching and during the transition from the histotrophic to the placental phase), day 13 (early placental phase), day 13.5–14.5 (during the late placental to early parturition phase), and 9–10 month post-partum, as well as non-pregnant control (Figure 1D; Lynch et al., 2015; Hansen et al., 2016; Griffith et al., 2017; Griffith et al., 2019). We found that HAND2 was abundantly expressed in the non-pregnant endometrium and down-regulated throughout the histotrophic phase, reaching a low (TPM < 2) at 12.5d, and subsequently increasing in expression in the 13.5-14d samples near term. ESR1, FGF2, FGF9, FGF18 and several WNT genes that stimulate proliferation of the luminal epithelia were abundantly expressed (Figure 1D and Figure 1—source data 7), consistent with persistent estrogen signaling during pregnancy (Renfree and Blanden, 2000). Both HAND2 and ESR1 decrease during gestation (Figure 1D), suggesting that the inhibition of estrogen signaling by HAND2 seen in Eutherians does not occur in opossum endometrium – if it did, one would expect ESR1 expression to increase as HAND2 expression decreased. Similarly, HAND2 is down-regulated during pregnancy in the tammar wallaby (Figure 1—figure supplement 1—source data 1, Figure 1—figure supplement 1). Immunohistochemistry (IHC) on opossum endometrial sections from day 12.5 pregnant endometrium stained strongly for estrogen receptor alpha (ESR1; ERα) phosphorylated at serine 118, a mark of transcriptionally active ERα (Kato et al., 1995), as well as phosphorylated ERK1/2, and MUC1 (Figure 1—figure supplement 2), which is also consistent with active estrogen signaling.
HAND2 is expressed in endometrial stromal fibroblast lineage cells
To determine which cell types at the human maternal-fetal interface express HAND2, we used previously generated single-cell RNA-Seq (scRNA-Seq) data from the first trimester decidua (Vento-Tormo et al., 2018). HAND2 expression was almost entirely restricted to cell populations in the endometrial stromal fibroblast lineage (see Materials and methods for cell type naming convention), with particularly high expression in ESF2s and DSCs (Figure 2A). Interestingly, while it is generally thought that ESFs are not present in the pregnant endometrium, previous studies have demonstrated that ESFs retain a presence in the endometrium from the first trimester all the way to term (Richards et al., 1995; Suryawanshi et al., 2018; Muñoz-Fernández et al., 2019; Sakabe et al., 2020).
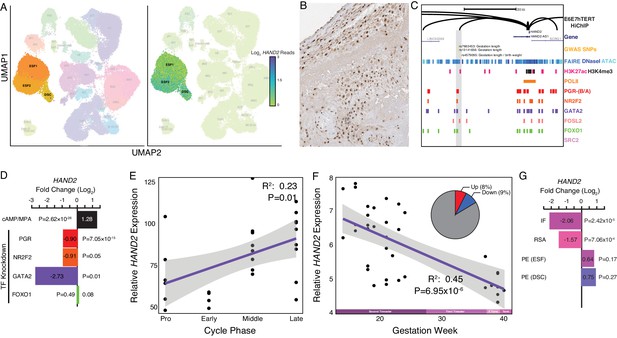
Expression of HAND2 at the maternal-fetal interface.
(A) UMAP clustering of scRNA-Seq data from human first trimester maternal-fetal interface. Left, clusters colored according to inferred cell type. The ESF1, ESF2, and DSC clusters are highlighted. Right, cells within clusters are colored according to HAND2 expression level. scRNA-Seq data from Vento-Tormo et al., 2018. (B) HAND2 protein expression in human pregnant decidua, with strong staining and localization in the nuclei of endometrial stromal cells. Image credit: Human Protein Atlas. (C) Regulatory landscape of the HAND2 locus. Chromatin loops inferred from H3K27ac HiChIP, regions of open chromatin inferred from FAIRE-, DNaseI, and ATAC-Seq, and the locations of histone modifications and transcription factor ChIP-Seq peaks are shown. The location of SNPs associated with gestation length / birth weight is also shown (highlighted in gray). Note that the HAND2 promoter forms a long-range loop to a region marked by H3K27ac and bound by PGR, NR2F2 (COUP-TFII), GATA2, FOSL2, and FOXO1. (D) HAND2 expression is up-regulated by in vitro decidualization of ESFs into DSC by cAMP/progesterone treatment, and down-regulated by siRNA-mediated knockdown of PGR and GATA2, but not NR2F2 or FOXO1. n = 3 per transcription factor knockdown. (E) Relative expression of HAND2 in the proliferative (n = 6), early (n = 4), middle (n = 9), and late (n = 8) secretory phases of the menstrual cycle. Note that outliers are excluded from the figure but not the regression; 95% CI is shown in gray. Gene expression data from Talbi et al., 2006. (F) Relative expression of HAND2 in the basal plate from mid-gestation to term (14–40 weeks, n = 36); 95% CI is shown in gray. Inset, percent of up- (Up) and down-regulated (Down) genes between weeks 14–19 and 37–40 of pregnancy (FDR ≤ 0.10). Gene expression data from Winn et al., 2007. (G) HAND2 expression is significantly down-regulated in the endometria of women with implantation failure (IF, n = 5) and recurrent spontaneous abortion (RSA, n = 5) compared to fertile controls (n = 5), but is not differentially expressed in ESFs or DSCs from women with preeclampsia (PE, n = 5) compared to healthy controls (n = 5). Gene expression data for RSA and IF from Lédée et al., 2011 and for PE from Garrido-Gomez et al., 2017.
© 2021, Human Protein Atlas. Figure 2B is adapted from the Human Protein Atlas (lower left part of top image cropped and rotated, image color auto adjusted). Published under a CC BY SA 3.0 unported license.
HAND2 protein was localized to nuclei in ESF lineage cells in human pregnant decidua (Figure 2B) from Human Protein Atlas IHC data (Uhlén et al., 2015). We also used existing functional genomics data to explore the regulatory status of the HAND2 locus (see Materials and methods for references). Consistent with active expression, the HAND2 locus in human DSCs is marked by histone modifications that typify enhancers (H3K27ac) and promoters (H3K4me3) and is located in a region of open chromatin as assessed by ATAC-, DNaseI- and FAIRE-Seq. Additionally, it is bound by transcription factors that establish endometrial stromal cell type identity and mediate decidualization, including the progesterone receptor (PGR), NR2F2 (COUP-TFII), GATA2, FOSL2, FOXO1, as well as polymerase II (Figure 2C). The HAND2 promoter loops to several distal enhancers, as assessed by H3K27ac HiChIP data generated from a normal hTERT-immortalized endometrial cell line (E6E7hTERT), including a region bound by PGR, NR2F2, GATA2, FOSL2, and FOXO1, that also contains SNPs associated with gestation length in recent GWAS (Warrington et al., 2019; Sakabe et al., 2020; Figure 2C). HAND2 was significantly upregulated by decidualization of human ESFs into DSCs by cAMP/progesterone treatment (Log2FC = 1.28, p = 2.62×10−26, FDR = 1.16×10−24), and significantly downregulated by siRNAs targeting PGR (Log2FC = −0.90, p = 7.05×10−15, FDR = 2.03×10−13) and GATA2 (Log2FC = −2.73, p = 0.01, FDR = 0.19) (Figure 2D). In contrast, siRNA-mediated knockdown of neither NR2F2 (Log2FC = −0.91, p = 0.05, FDR = 1.0) nor FOXO1 (Log2FC = 0.08, p = 0.49, FDR = 0.74) significantly altered HAND2 expression (Figure 2D and see Materials and methods for references).
Differential HAND2 expression throughout the menstrual cycle and pregnancy
Our observation that HAND2 is progesterone responsive suggests it may be differentially expressed throughout the menstrual cycle and pregnancy. To explore this possibility, we utilized previously published gene expression datasets generated from the endometrium across the menstrual cycle (Talbi et al., 2006) and from the basal plate from mid-gestation to term (Winn et al., 2007). HAND2 expression tended to increase from proliferative through the early and middle secretory phases, reaching a peak in the late secretory phase of the menstrual cycle (Figure 2E). In stark contrast, HAND2 decreases in expression from the first trimester to term (Figure 2F). For comparison, 9% of genes were down-regulated between weeks 14–19 and 37–40 of pregnancy (FDR ≤ 0.10). We also used previously published gene expression datasets to explore if HAND2 was associated with disorders of pregnancy (Lédée et al., 2011; Garrido-Gomez et al., 2017). Although sample sizes of these datasets are small, and the intrinsic temporo-spatial heterogeneity of the endometrium remains a potential confounding factor, we found that HAND2 was dysregulated in the endometria of women with implantation failure (IF) and recurrent spontaneous abortion (RSA), while it was not differentially expressed in ESFs or DSCs from women with preeclampsia (PE), compared to controls (Figure 2G).
HAND2 regulates a distinct set of target genes
HAND2 expression has previously been shown to play a role in orchestrating the transcriptional response to progesterone during decidualization in human and mouse DSCs (Huyen and Bany, 2011; Li et al., 2011; McConaha et al., 2011; Shindoh et al., 2014; Murata et al., 2020). However, whether HAND2 has functions in other endometrial stromal lineage cells such as ESFs, which persist in the pregnant endometrium till term (Richards et al., 1995; Suryawanshi et al., 2018; Muñoz-Fernández et al., 2019; Sakabe et al., 2020) but have received less attention than DSCs during pregnancy, is unknown. Therefore, we used siRNA to knockdown HAND2 expression in human hTERT-immortalized ESFs (T-HESC) and assayed global gene expression changes by RNA-Seq 48 hr after knockdown. We found that HAND2 was knocked down ~78% (p = 7.79×10−3) by siRNA treatment (Figure 3A), which dysregulated the expression of 553 transcripts (489 genes) at FDR ≤ 0.10 (Figure 3A and Figure 3—source data 1). Genes dysregulated by HAND2 knockdown were enriched in several pathways and human phenotype ontologies relevant to endometrial stromal cells and pregnancy in general (Figure 3B and Figure 3—source data 2, 3).
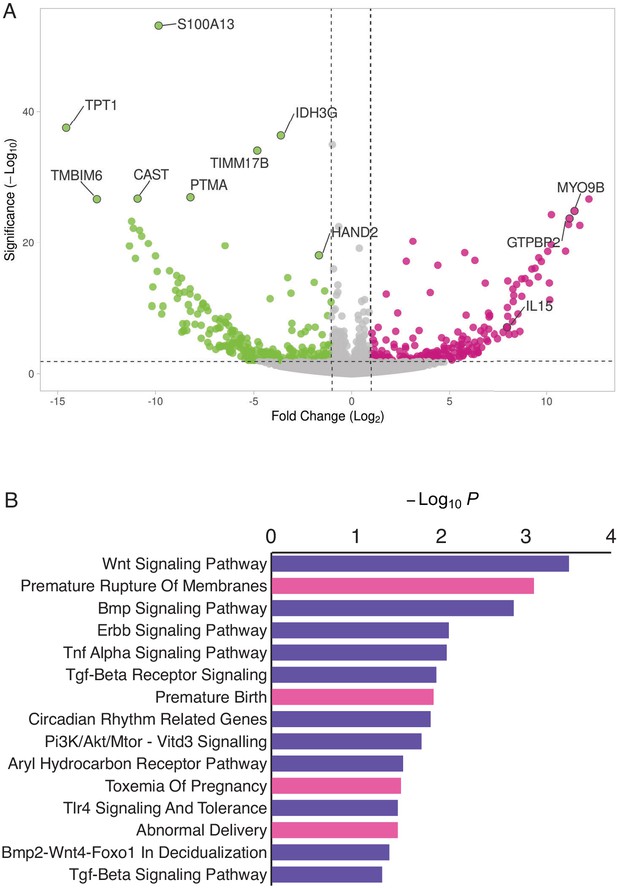
HAND2 regulates a distinct set of target genes, including IL15.
(A) Volcano plot of gene expression upon HAND2 knockdown. Only genes that are significantly differentially expressed (DE) with FDR ≤ 0.10 are colored. Genes with ≥ 1 fold changes in expression are shown in pink (up-regulated), green (down-regulated) or gray (not differentially expressed). X-axis shows log2 fold change, Y-axis shows Wald statistic p-value, horizontal dashed line indicates FDR = 0.10. Full list of DE genes can be found in Figure 3—source data 1. (B) Pathways (purple) and human phenotype ontologies (pink) in which genes dysregulated upon HAND2 knockdown are enriched. We used a hypergeometric p-value to determine enriched pathway and disease ontology terms. The Benjamini-Hochburg Adjusted p-value (FDR), Odds Ratio, Combined Score, and Genes associated with each term can be found in Figure 3—source data 2 and Figure 3—source data 3.
-
Figure 3—source data 1
Genes differentially expressed (DE) upon HAND2 knockdown.
RNA-Seq reads were mapped to hg38 using HISAT2, transcripts were assembled and quantified using StringTie, and DE genes were identified using DESeq2. The reference file for StringTie guided assembly was wgEncodeGencodeBasicV33. Columns indicate: Ensembl stable transcript IDs (A), HUGO gene names (B), Base mean expression level (C), Log2 fold change upon HAND2 knockdown (D), Standard Error of Log2 fold change (E), Walds-Statistic for differential expression (F), p-value of the Walds-Statitic (G), and Benjamini-Hochberg corrected p-values (H).
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig3-data1-v2.xlsx
-
Figure 3—source data 2
Pathways (Wikipathway 2019) enriched among genes differentially expressed by HAND2 knockdown.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig3-data2-v2.xlsx
-
Figure 3—source data 3
Human phenotype (disease) ontology terms enriched among genes differentially expressed by HAND2 knockdown.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig3-data3-v2.xlsx
Enriched pathways play a role in decidualization (e.g. ‘Wnt Signaling’, ‘BMP Signaling’, ‘ErbB Signaling’, ‘TGF-beta Receptor Signaling’ and ‘BMP2-WNT4-FOXO1 Pathway in Human Primary Endometrial Stromal Cell Differentiation’), as well as in placental bed development disorders and preeclampsia, the induction of pro-inflammatory factors via nuclear factor-κB (NFκB), mediation of maternal immunotolerance to the fetal allograft, circadian rhythm in association with implantation and parturition, and the decidual inflammation, senescence, and parturition. Selected pathways and associated references are listed in Table 1.
Genes dysregulated by HAND2 knockdown are enriched in pathways relevant to endometrial stromal cells and pregnancy in general.
| Enriched pathway | Roles in ESFs and pregnancy | References |
|---|---|---|
| Wnt Signaling | Decidualization | Peng et al., 2008; Hayashi et al., 2009; Sonderegger et al., 2010; Franco et al., 2011; Wang et al., 2013 |
| BMP Signaling | Decidualization | Ying and Zhao, 2000; Lee et al., 2007; Li et al., 2007; Wetendorf and DeMayo, 2012 |
| ErbB Signaling | Decidualization | Lim et al., 1997; Klonisch et al., 2001; Large et al., 2014 |
| TGF-beta Receptor Signaling | Decidualization | Jones et al., 2006; Li, 2014; Ni and Li, 2017 |
| BMP2-WNT4-FOXO1 Pathway in Human Primary Endometrial Stromal Cell Differentiation | Decidualization | Gellersen and Brosens, 2003; Buzzio et al., 2006; Lee et al., 2007; Li et al., 2007; Brayer et al., 2011; Lynch et al., 2009; Kajihara et al., 2013 |
| AGE/RAGE Pathway | Placental bed development disorders Preeclampsia Induction of pro-inflammatory factors via nuclear factor-κB (NFκB) | Chekir et al., 2006; Lappas et al., 2007; Oliver et al., 2011; Guedes-Martins et al., 2013 |
| Aryl Hydrocarbon Receptor Pathway | Mediation of maternal immunotolerance to fetal allograft | Munn et al., 1998; Abbott et al., 1999; Funeshima et al., 2005; Hao et al., 2013 |
| Circadian Rhythm Related Genes | Implantation and parturition | Roizen et al., 2007; Olcese, 2012; Olcese et al., 2013; Greenhill, 2014; Menon et al., 2016 |
| RAC1/PAK1/p38/MMP2 Pathway | Decidual inflammation, senescence and parturition | Menon et al., 2016 |
Enriched human phenotype ontology terms were related to complications of pregnancy, including ‘Premature Rupture of Membranes’, ‘Premature Birth’, ‘Toxemia of Pregnancy’ (preeclampsia) and ‘Abnormal Delivery’. We also observed that several genes in the NFκB pathway, such as MYD88, CHUK, IκBKE, NFκBIE, and RTKN2 were differentially expressed; NFκB signaling has been associated with the molecular etiology of preterm birth (Allport et al., 2001; Lindström and Bennett, 2005).
HAND2 regulates IL15 expression in endometrial stromal fibroblast lineage cells
Among the genes dysregulated by HAND2 knockdown in ESFs was IL15 (Log2FC = 7.98, p = 7.91×10−8, FDR = 1.49×10−5), a pleiotropic cytokine previously shown to be expressed in the endometrium and decidua (Figure 3A and Table 2; Kitaya et al., 2000; Okada et al., 2000; Dunn et al., 2002; Okada et al., 2004; Godbole and Modi, 2010). IL15 was robustly expressed at the first trimester maternal-fetal interface in stromal fibroblast lineage cells (Figure 4A), and there was a general correlation between HAND2 and IL15 expression in single cells (Figure 4A inset) (Vento-Tormo et al., 2018). IL15 protein localized to cytoplasm in ESF lineage cells in human pregnant decidua (Figure 4B) in Human Protein Atlas IHC data. The IL15 promoter loops to several distal sites in H3K27ac HiChIP data from E6E7hTERT endometrial cells including to regions bound by PGR, NR2F2, GATA2, FOSL2, FOXO1, and SRC2, an intrinsic histone acetyltransferase that is a transcriptional co-factor of ligand-dependent hormone receptors (Figure 4C; see Materials and methods for references). The IL15 promoter also loops to a putative enhancer in its first intron that contains a PGR-binding site and SNPs marginally associated with a maternal effect on offspring birth weight (rs190663174, p = 6×10−4) by GWAS (Warrington et al., 2019). IL15 was significantly upregulated by in vitro decidualization of human ESFs into DSCs by cAMP/progesterone treatment (Log2FC = 2.15, p = 2.58×10−33, FDR = 1.59×10−31), and significantly downregulated by siRNAs targeting PGR (Log2FC = −1.24, p = 6.23×10−15, FDR = 1.80×10−13) and GATA2 (Log2FC = −2.08, p = 4.16×10−3, FDR = 0.14) (Figure 4D), but not NR2F2 (Log2FC = 0.19, p = 0.38, FDR = 0.93) or FOXO1 (Log2FC = 0.29, p = 0.04, FDR = 0.22) (Figure 4D; see Materials and methods for references). Although HAND2 binding data is not available for human stromal fibroblast lineage cells, several HAND2 binding motifs (≥0.85 motif match) are located within enhancers that loop to the IL15 promoter.
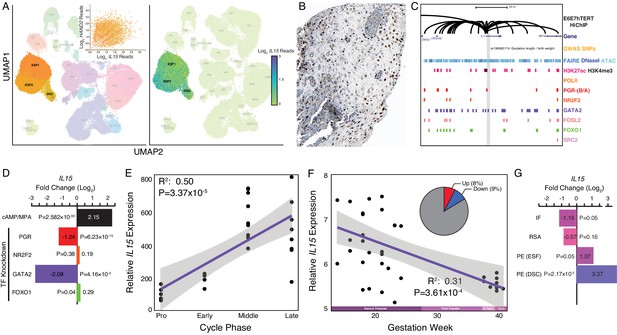
Expression of IL15 at the maternal-fetal interface.
(A) UMAP clustering of scRNA-Seq data from human first trimester maternal-fetal interface. Left, clusters colored according to inferred cell type. The ESF1, ESF2, and DSC clusters are highlighted. Inset, per cell expression of HAND2 and IL15 in ESF1s, ESF2s, and DSCs. Right, cells within clusters are colored according to IL15 expression level. scRNA-Seq data from Vento-Tormo et al., 2018. (B) IL15 protein expression in human pregnant decidua, with strong cytoplasmic staining in endometrial stromal cells. Image credit: Human Protein Atlas. (C) Regulatory landscape of the IL15 locus. Chromatin loops inferred from H3K27ac HiChIP, regions of open chromatin inferred from FAIRE-, DNaseI, and ATAC-Seq, and the locations of histone modifications and transcription factor ChIP-Seq peaks are shown. The location of an SNP associated with gestation length / birth weight is also shown (highlighted in gray). Note that the IL15 promoter forms many long-range loops to regions marked by H3K27ac and bound by PGR, NR2F2 (COUP-TFII), GATA2, FOSL2, FOXO1, and SRC2. (D) IL15 expression is upregulated by in vitro decidualization of ESFs into DSC by cAMP/progesterone treatment, and downregulated by siRNA-mediated knockdown of PGR and GATA2 but not NR2F2 or FOXO1. n = 3 per transcription factor knockdown. (E) Relative expression of IL15 in the proliferative (n = 6), early (n = 4), middle (n = 9), and late (n = 8) secretory phases of the menstrual cycle. Note that outliers are excluded from the figure but not the regression; 95% CI is shown in gray. Gene expression data from Talbi et al., 2006. (F) Relative expression of IL15 in the basal plate from mid-gestation to term (14–40 weeks, n = 36); 95% CI is shown in gray. Inset, percent of up- (Up) and downregulated (Down) genes between weeks 14–19 and 37–40 of pregnancy (FDR ≤ 0.10). Gene expression data from Winn et al., 2007. (G) IL15 expression is significantly upregulated in DSCs from women with preeclampsia (PE, n = 5) compared to healthy controls (n = 5), while it is only marginally upregulated in ESFs from the same patient group. It is also marginally downregulated in the endometria of women with implantation failure (IF, n = 5) and it is not differentially expressed in the endometria of women with recurrent spontaneous abortion (RSA, n = 5) compared to fertile controls (n = 5). Gene expression data for RSA and IF from Lédée et al., 2011 and for PE from Garrido-Gomez et al., 2017.
© 2021, Human Protein Atlas. Figure 4B is adapted from the Human Protein Atlas (top image cropped, image color auto adjusted). Published under a CC BY SA 3.0 unported license.
Exemplar genes differentially expressed in ESFs upon siRNA-mediated HAND2 knockdown.
Mean, base mean expression level. FC, log2 fold change. SE, standard error in log2 fold change. WS, Wald statistic. p-Value, Wald test p-value. Adj-p, Benjamini-Hochberg (BH) adjusted p-value. Function, function of gene inferred from Wikipathway 2019 human annotation. Association with preterm birth (PTB, HP:0001622) and premature rupture of membranes (PROM, HP:0001788) inferred from human phenotype ontology annotation*.
| Gene | Mean | FC | SE | WS | p-Value | Adj-p | Function |
|---|---|---|---|---|---|---|---|
| ARNT2 | 44.47 | 1.20 | 0.28 | 4.30 | 1.73E-05 | 1.89E-03 | AHR signaling / Circadian rhythm |
| ARNTL | 42.52 | −2.95 | 0.80 | −3.67 | 2.46E-04 | 1.79E-02 | AHR signaling / Circadian rhythm |
| IL15 | 63.31 | 7.98 | 1.49 | 5.37 | 7.91E-08 | 1.49E-05 | Cell migration |
| BMP4 | 58.10 | −8.34 | 1.25 | −6.66 | 2.75E-11 | 8.28E-09 | Decidualization |
| GSK3B | 69.23 | −5.53 | 1.48 | −3.73 | 1.94E-04 | 1.49E-02 | Decidualization |
| HAND2 | 136.06 | −1.63 | 0.18 | −8.85 | 9.10E-19 | 7.64E-16 | Knocked down gene |
| CHUK | 765.77 | 0.44 | 0.14 | 3.14 | 1.67E-03 | 7.94E-02 | NFκB pathway |
| IκBKE | 9.91 | −5.41 | 1.65 | −3.27 | 1.06E-03 | 5.72E-02 | NFκB pathway |
| MYD88 | 95.02 | −0.93 | 0.26 | −3.55 | 3.88E-04 | 2.59E-02 | NFκB pathway |
| NFκBIE | 106.63 | −0.78 | 0.25 | −3.08 | 2.10E-03 | 9.53E-02 | NFκB pathway |
| RTKN2 | 50.64 | 8.02 | 1.33 | 6.02 | 1.75E-09 | 4.05E-07 | NFκB pathway |
| CRKL | 1278.85 | −0.31 | 0.09 | −3.31 | 9.21E-04 | 5.10E-02 | PTB |
| EOGT | 233.65 | −2.77 | 0.80 | −3.45 | 5.63E-04 | 3.38E-02 | PTB |
| LMNA | 773.24 | 6.97 | 1.57 | 4.44 | 8.92E-06 | 1.06E-03 | PTB; PROM |
| PEX11B | 181.31 | 10.05 | 1.12 | 9.00 | 2.28E-19 | 2.06E-16 | PTB |
| SERPINH1 | 1654.90 | −6.01 | 1.31 | −4.59 | 4.40E-06 | 5.61E-04 | PROM |
| SLC17A5 | 826.28 | 0.76 | 0.13 | 5.81 | 6.34E-09 | 1.40E-06 | PTB |
| ZMPSTE24 | 2140.67 | −0.19 | 0.05 | −4.10 | 4.10E-05 | 3.92E-03 | PTB; PROM |
-
*Extended list of genes associated with the pathologies of pregnancy and their expression levels can be found in Table 2—source data 1 used to generate Figures 2G and 4G. (GSE26787 = recurrent spontaneous abortion [RSA] and implantation failure [IF]; GSE91077 = ESFs and DSCs from women with preeclampsia [PE]).
-
Table 2—source data 1
Differential expression of genes in RSA, IF and PE (in ESFs and DSCs) in relation to genes differentially expressed upon HAND2 knockdown in ESFs.
- https://cdn.elifesciences.org/articles/61257/elife-61257-table2-data1-v2.xlsx
Differential IL15 expression throughout the menstrual cycle and pregnancy
Our observations that HAND2 is progesterone responsive and differentially expressed throughout the menstrual cycle and pregnancy suggest that IL15, which is controlled by HAND2 as well as cAMP/progesterone/PGR/GATA2, may be similarly regulated. Indeed, like HAND2, we found that IL15 expression increased as the menstrual cycle progressed, peaking in the middle-late secretory phases (Figure 4E; Talbi et al., 2006) and decreased in expression from the first trimester to term (Figure 4F; Winn et al., 2007). IL15 expression was also dysregulated in the endometria of women with implantation failure but not recurrent spontaneous abortion, compared to fertile controls (Figure 4G; Lédée et al., 2011). In women with preeclampsia, IL15 was not dysregulated in ESFs, but it was expressed significantly higher in DSCs, compared to controls (Figure 4G; Garrido-Gomez et al., 2017). Thus, like HAND2, IL15 is differentially expressed throughout the menstrual cycle and pregnancy, and in the endometria of women with implantation failure.
ESF-derived IL15 promotes NK and trophoblast migration
Endometrial stromal cells promote the migration of uterine natural killer (uNK) (Chen et al., 2011) and trophoblast cells (Graham and Lala, 1991; Paiva et al., 2009; Zhu et al., 2009; Godbole et al., 2011). IL15, in particular, stimulates the migration of uNK cells (Allavena et al., 1997; Verma et al., 2000; Ashkar et al., 2003; Barber and Pollard, 2003; Kitaya et al., 2005) and the human choriocarcinoma cell line, JEG-3 (Zygmunt et al., 1998). Therefore, we tested whether ESF-derived IL15 influenced the migration of primary human NK cells and immortalized first trimester extravillous trophoblasts (HTR-8/SVneo) in trans-well migration assays (Figure 5A). We found that ESF media supplemented with recombinant human IL15 (rhIL15) was sufficient to stimulate the migration of NK and HTR-8/SVneo cells to the lower chamber of trans-wells, compared to non-supplemented media (Figure 5B,C and Figure 5—source data 1, 2). Conditioned media from ESFs with siRNA-mediated HAND2 knockdown increased migration of both NK and HTR-8/SVneo compared to negative control (i.e. non-targeting siRNA; Figure 5B,C). Conditioned media from ESFs with siRNA-mediated IL15 knockdown reduced migration of both NK and HTR-8/SVneo cells compared to negative control (Figure 5B,C). Similarly, ESF conditioned media supplemented with anti-IL15 antibody reduced cell migration compared to media supplemented with control IgG antibody (Figure 5B,C).
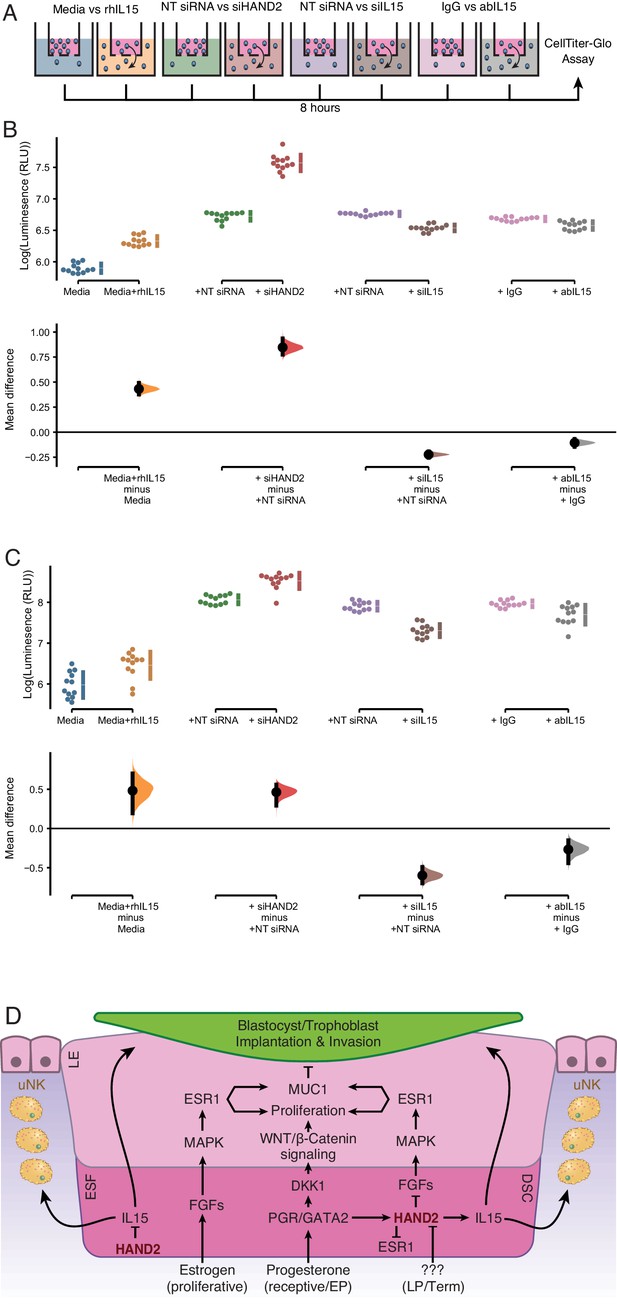
ESF-derived IL15 promotes NK and trophoblast migration in trans-well assays.
(A) Cartoon of trans-well migration assay comparisons. Cells that migrated to the lower chamber were quantified using the CellTiter-Glo luminescent cell viability assay after 8 hr. (B) Primary natural killer (NK) cells. Raw luminescence data (RLU) from cells in the lower chamber is shown in the upper panel, mean difference (effect size) in experiment minus control luminescence values are shown as dots with the 95% confidence interval indicated by vertical bars in the lower panel; distribution estimated from 5000 bootstrap replicates. The mean difference between Media and media supplemented with recombinant human IL15 (Media+rhIL15) is 0.432 [95.0% CI: 0.376–0.492]; p = 0.00. The mean difference between ESFs transiently transfected with non-targeting siRNA (NT siRNA) and HAND2-specific siRNAs (siHAND2) is 0.847 [95.0% CI: 0.774–0.936]; p = 0.00. The mean difference between ESFs transiently transfected with NT siRNA and IL15-specific siRNAs (siIL15) is −0.223 [95.0% CI: −0.256 – −0.192]; p = 0.00. The mean difference between ESF media neutralized with a non-specific antibody (IgG) or IL15-specific antibody (abIL15) is −0.106 [95.0% CI: −0.147 – −0.067]; p = 0.00. n = 12. (C) Extravillous trophoblast cell line HTR-8/SVneo. Raw luminescence data (RLU) from cells in the lower chamber is shown in the upper panel, mean difference (effect size) in experiment minus control luminescence values are shown as dots with the 95% confidence interval indicated by vertical bars in the lower panel; distribution estimated from 5000 bootstrap replicates. The mean difference between Media and Media+rhIL15 is 0.482 [95.0% CI: 0.193–0.701]; p = 0.002. The mean difference between ESFs transiently transfected with NT siRNA and siHAND2 is 0.463 [95.0% CI: 0.291–0.559]; p = 0.00. The mean difference between ESFs transiently transfected with NT siRNA and siIL15 is −0.598 [95.0% CI: −0.698–0.490]; p = 0.00. The mean difference between ESF media neutralized with IgG or abIL15 is −0.267 [95.0% CI: −0.442 – −0.151]; p = 0.0004. n = 12. (D) Model of HAND2 functions in the endometrium. During the proliferative phase HAND2 inhibits IL15, and thus the migration of uNK and trophoblast cells. In the receptive phase, HAND2 activates IL15, which promotes migration of uNK and trophoblast cells. In the receptive phase and early pregnancy (EP), HAND2 suppresses estrogen signaling by down-regulating FGFs and directly binding and inhibiting the ligand-dependent transcriptional activation function of ESR1. During late pregnancy/term (LP/Term), reduced HAND2 expression mitigates its anti-estrogenic functions. Parturition signal unknown (???). ESF = endometrial stromal fibroblasts (proliferative phase), DSC = decidual stromal cells (receptive phase), LE = luminal epithelium.
-
Figure 5—source data 1
Raw and log transformed luminescence data for NK trans-well migration assays.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig5-data1-v2.xlsx
-
Figure 5—source data 2
Raw and log transformed luminescence data for HTR-8 trans-well migration assays.
- https://cdn.elifesciences.org/articles/61257/elife-61257-fig5-data2-v2.xlsx
Discussion
Eutherian mammals evolved a suite of traits that support pregnancy, including an interrupted estrous cycle allowing for prolonged gestation lengths, maternal-fetal communication, implantation, maternal immunotolerance and recognition of pregnancy, and thus are uniquely afflicted by disorders of these processes. When searching for clues as to how variation in normal physiological functions can lead to dysfunction and disease, deeper understanding of the evolutionary and developmental histories of organ and tissues has the potential to provide novel insights. Here, we used evolutionary transcriptomics to identify genes that evolved to be expressed on the maternal side (endometrium) of the maternal-fetal interface during the origins of pregnancy in Eutherians, and hence may also contribute to pregnancy complications such as infertility, recurrent spontaneous abortion, and preterm birth.
Among the genes recruited into endometrial expression in Eutherians, we identified HAND2, a pleiotropic transcription factor that plays an essential role in suppressing estrogen signaling at the time of uterine receptivity to blastocyst embedding, through its down-regulation of pro-estrogenic genes and by directly inhibiting the transcriptional activities of the estrogen receptor (Huyen and Bany, 2011; Li et al., 2011; Shindoh et al., 2014; Fukuda et al., 2015; Mestre-Citrinovitz et al., 2015; Murata et al., 2019). Consistent with these functions, we found evidence of estrogen activity in the endometrium of pregnant opossum. Earlier research detected similar activity in the gravid oviduct of birds and reptiles (Means et al., 1975; Kato et al., 1992; Girling, 2002; González-Morán, 2015). These data indicate that suppression of estrogen signaling during the window of uterine receptivity to implantation is an evolutionary innovation of Eutherian mammals, which involved the recruitment of HAND2 and its anti-estrogenic functions into endometrial expression.
The roles of HAND2 in DSCs and implantation are well-understood (Huyen and Bany, 2011; Li et al., 2011; Shindoh et al., 2014; Fukuda et al., 2015; Mestre-Citrinovitz et al., 2015; Murata et al., 2019; Šućurović et al., 2020). In contrast, the function(s) of HAND2 at other stages of pregnancy and in ESFs remain relatively unexplored, despite the persistence of ESFs in the pregnant endometrium until term (Richards et al., 1995; Suryawanshi et al., 2018; Muñoz-Fernández et al., 2019; Sakabe et al., 2020). HAND2, for example, plays a role in orchestrating the transcriptional response to progesterone during decidualization (Huyen and Bany, 2011; Li et al., 2011; McConaha et al., 2011; Shindoh et al., 2014; Murata et al., 2020). Similarly, Hand2 knockout mice are infertile because of persistent estrogen signaling during the window of implantation, leading to implantation failure (Li et al., 2011; Jones et al., 2013). The functions of Hand2 at other stages of pregnancy could unfortunately not be explored in these Hand2 knockout mice as the conditional targeting strategy knocks out Hand2 in Pgr-expressing cells. Hand2 expression is thereby eliminated upon initiation of Pgr expression in the uterus, coincident with the onset of sexual maturity.
We knocked down HAND2 in ESFs and found that downstream dysregulated genes were enriched for human phenotype ontologies related to disorders of pregnancy, including ‘Premature Rupture of Membranes’, ‘Premature Birth’, and ‘Abnormal Delivery’, suggesting that HAND2 has functions throughout pregnancy and in parturition. Indeed, we discovered that SNPs recently implicated in the regulation of gestation length and birth weight by GWAS (Warrington et al., 2019; Sakabe et al., 2020) make long-range interactions to the HAND2 promoter. Also of note, HAND2 expression is significantly higher in placental villous samples from idiopathic spontaneous preterm birth (isPTB) compared to term controls (Brockway et al., 2019). However, this difference may be related to gestational age rather than the etiology of PTB (Eidem et al., 2016).
Additionally, analyzing previously published datasets we noticed that HAND2 expression decreases throughout gestation. Unlike the majority of Eutherians, where parturition closely follows the significant drop in progesterone concentrations in maternal peripheral blood, this is not the case for humans and other Old World primates (Ratajczak et al., 2010), where progesterone levels keep rising throughout gestation, reaching maximum at birth. These observations, combined with the central role of HAND2 in mediating the anti-estrogenic actions of progesterone, suggest decreased HAND2 at the end of pregnancy may contribute to the estrogen-dominant uterine environment at the onset of labor (Pinto et al., 1966; Pepe and Albrecht, 1995; Mesiano et al., 2002; Smith et al., 2009a; Ratajczak et al., 2010; Welsh et al., 2012), despite high systemic progesterone. Low HAND2 at the end of pregnancy in humans is therefore most likely not directly related to progesterone concentrations, suggesting that an unidentified inhibitory signal reduces endometrial HAND2 expression. While taken collectively these data indicate a role for HAND2 in pre/term birth, a direct mechanistic link between HAND2 expression and the timing of parturition remains to be demonstrated.
One of the genes dysregulated by HAND2 knockdown was the multifunctional cytokine IL15, which plays important roles in innate and adaptive immunity. In the context of pregnancy, it is important for the recruitment of uterine natural killer (uNK) cells to the endometrium (Kitaya et al., 2000; Verma et al., 2000; Ashkar et al., 2003; Barber and Pollard, 2003; Kitaya et al., 2005; Laskarin et al., 2006). The roles of uNK cells in the remodeling of uterine spiral arteries and regulating trophoblast invasion are well known (Zygmunt et al., 1998; Hanna et al., 2006; Smith et al., 2009b; Burke et al., 2010; Hazan et al., 2010; Lash et al., 2010; Bany et al., 2012; Robson et al., 2012; Zhang et al., 2013; Lima et al., 2014; Fraser et al., 2015; Felker and Croy, 2016; Renaud et al., 2017). Endometrial stromal cell-derived IL15 is also necessary for the selective targeting and clearance of senescent endometrial stromal cells from the implantation site by uNK cells, which is essential for endometrial rejuvenation and remodeling at embryo implantation (Brighton et al., 2017). Dysregulation of uNK cell-mediated clearance of these senescent cells has also been implicated in recurrent pregnancy loss (Lucas et al., 2020). We found that, like HAND2, IL15 decreases throughout gestation and both genes increase in expression as the menstrual cycle progresses.
Unexpectedly, however, while previous studies showed that HAND2 induces IL15 in DSCs (Shindoh et al., 2014; Murata et al., 2020), we discovered that HAND2 inhibited IL15 expression in ESFs, indicating a switch in regulatory activity sometime during early menstrual cycle. These data suggest that HAND2 regulates the appropriate timing of endometrial IL15 expression during the menstrual cycle and throughout pregnancy, and thus the appropriate timing of uNK cell recruitment, trophoblast migration and the clearance of senescent endometrial stromal cells from the implantation site (Figure 5D). While the signals that initiate parturition in humans and other Catarrhine primates are unknown, it has been proposed that a ‘decidual clock’ may regulate the successful establishment and maintenance of pregnancy, such that severe decidualization defects lead to infertility, moderate defects lead to recurrent pregnancy loss/recurrent spontaneous abortion, and mild defects lead to preterm birth (Norwitz et al., 2015). Our results suggest that part of this clock may be the transition of ESFs that persist in the endometrium during pregnancy to DSCs, and/or DSCs into senescent DSCs (snDSCs) which no longer express IL15 (Lucas et al., 2020), leading to a shift in the balance of tolerizing immune cells at the maternal-fetal interface and thus withdrawal of maternal immunotolerance of the fetal allograft. Thus, our observation of low HAND2 and IL15 near term may reflect a reduction of anti-inflammatory DSCs (high HAND2 and IL15) and an accumulation of pro-inflammatory snDSCs (low HAND2 and IL15) at the maternal-fetal interface. Additional work is needed to elucidate the mechanisms that underlie change in HAND2-IL15 dynamics and determine whether these progressive cell state changes occur during gestation.
Decreased HAND2 and IL15 expression near term and their influence on immune cells at the maternal-fetal interface may also play a role in parturition. Pre/term labor is known to be associated with elevated inflammation and an influx of immune cells into utero-placental tissues (Thomson et al., 1999; Young et al., 2002; Osman et al., 2003; Gomez-Lopez et al., 2010; Rinaldi et al., 2011; Rinaldi et al., 2015; Hamilton et al., 2012; Shynlova et al., 2013; Bartmann et al., 2014; Menon et al., 2016; Peters et al., 2016; Wilson and Mesiano, 2020). uNK cells are abundant throughout gestation (Bulmer et al., 1991; King et al., 1991; Moffett-King, 2002; Williams et al., 2009; Bartmann et al., 2014), but whether they play a role in late pregnancy and parturition is unclear. However, depletion of uNK cells rescues LPS-induced preterm birth in IL10-null mice (Murphy et al., 2005), indicating they contribute to infection/inflammation-induced preterm parturition (Murphy et al., 2009). CD16+CD56dim (cytotoxic) uNK cells have also been observed in the decidua and the placental villi of women with preterm but not term labor, suggesting an association between dysregulation of uNK cells and preterm birth in humans (Gomaa et al., 2017). uNK cells are associated with other pregnancy complications in humans such as fetal growth restriction, preeclampsia, and recurrent spontaneous abortion (Moffett et al., 2004; Hiby et al., 2010; Wallace et al., 2013; Wallace et al., 2014; Kieckbusch et al., 2014). Taken together, these data indicate that uNK cells may act downstream of HAND2-IL15 signaling in the timing of parturition.
Conclusions
Here, we show that HAND2 evolved to be expressed in endometrial stromal cells in the Eutherian stem-lineage, coincident with the evolution of suppressed estrogen signaling during the window of implantation and an interrupted reproductive cycle during pregnancy, which necessitated a means to regulate the length of gestation. Our data suggest that HAND2 may contribute to the regulation of gestation length by promoting an estrogen dominant uterine environment near term and through its effect on IL15 signaling and uNK cell function. To further expand our understanding of HAND2 functions at the molecular mechanistic level, multiple technical and ethical difficulties associated with studying human pregnancy in vivo will need to be overcome. Therefore, recently developed organoid models of the human maternal-fetal interface (Rinehart et al., 1988; Boretto et al., 2017; Turco et al., 2017; Turco et al., 2018; Marinić et al., 2020), which allow for in vitro 3D manipulation, will be instrumental.
Materials and methods
Endometrial gene expression profiling and ancestral transcriptome reconstruction
Data collection
Request a detailed protocolWe obtained previously generated RNA-Seq data from endometria of amniotes by searching NCBI BioSample, Sequence Read Archive (SRA), and Gene Expression Omnibus (GEO) databases for anatomical terms referring to the portion of the female reproductive tract (FRT), including ‘uterus’, ‘endometrium’, ‘decidua’, ‘oviduct’, and ‘shell gland’, followed by manual curation to identify those datasets that included the FRT region specialized for maternal-fetal interaction or shell formation. Datasets that did not indicate whether samples were from pregnant or gravid females were excluded, as were those composed of multiple tissue types. Species included in this study and their associated RNA-Seq accession numbers are included in Figure 1—source data 1.
New RNA-Seq data
Request a detailed protocolEndometrial tissue samples from the pregnant uteri of baboon (n = 3), mouse (n = 3), hamster (n = 3), bat (n = 2), and squirrel (n = 2) were dissected and mailed to the University of Chicago in RNA-Later. These samples were further dissected to remove myometrium, luminal epithelium, and extra-embryonic tissues, and then washed three times in ice cold PBS to remove unattached cell debris and red blood cells. Total RNA was extracted from the remaining tissue using the RNeasy Plus Mini Kit (74134, QIAGEN) per manufacturer’s instructions. RNA concentrations were determined by Nanodrop 2000 (Thermo Scientific). A total amount of 2.5 μg of total RNA per sample was submitted to the University of Chicago Genomics Facility for Illumina Next Gen RNA sequencing. Quality was assessed with the Bioanalyzer 2100 (Agilent). A total RNA library was generated using the TruSEQ stranded mRNA with RiboZero depletion (Illumina) for each sample. The samples were fitted with one of six different adapters with a different 6-base barcode for multiplexing. Completed libraries were run on an Illumina HiSEQ2500 with v4 chemistry on two replicate lanes for hamster and one lane for other species of an eight lane flow cell, generating 30–50 million 50 bp single-end reads per sample.
Multispecies RNA-Seq analysis
Request a detailed protocolFor all RNA-Seq analyses, we used Kallisto (Bray et al., 2016) version 0.42.4 to pseudo-align the raw RNA-Seq reads to reference transcriptomes (see Figure 1—source data 1 for reference genome assemblies) and to generate transcript abundance estimates. We used default parameters bias correction, and 100 bootstrap replicates. Kallisto outputs consist of transcript abundance estimates in Transcripts Per Million (TPM), which were used to determine gene expression.
Ancestral transcriptome reconstruction
Request a detailed protocolWe previously showed that genes with TPM ≥ 2.0 are actively transcribed in endometrium while genes with TPM < 2.0 lack hallmarks of active transcription such as promoters marked with H3K4me3 (Wagner et al., 2012; Wagner et al., 2013). Based on these findings, we transformed values of transcript abundance estimates into discrete character states, such that genes with TPM ≥ 2.0 were coded as expressed (state = 1), genes with TPM < 2.0 were coded as not expressed (state = 0), and genes without data in specific species coded as missing (state = ?). The binary encoded endometrial gene expression dataset generally grouped species by phylogenetic relatedness, suggesting greater signal-to-noise ratio than raw transcript abundance estimates. Therefore, we used the binary encoded endometrial transcriptome dataset to reconstruct ancestral gene expression states and trace the evolution of gene expression gains (0 → 1) and losses (1 → 0) in the endometria across vertebrate phylogeny (Figure 1A). We used Mesquite (Maddison and Maddison, 2019) (v3.02) with parsimony optimization to reconstruct ancestral gene expression states, and identify genes that gained or lost endometrial expression. Expression was classified as an unambiguous gain if a gene was not inferred as expressed at a particular ancestral node (state = 0) but inferred as expressed (state = 1) in a descendent of that node, and vice versa for the classification of a loss of endometrial expression (Figure 1—source data 2). Parsimony optimization of ancestral states results in three ancestral state reconstructions (ASRs): ‘ambiguous’, ‘most-parsimonious’, and ‘unambiguous’. An ambiguous ASR is not resolved and interpreted as unknown, a most-parsimonious ASR is ‘potentially’ the ASR but not necessarily so because other ancestral states are also possible, while an unambiguous ASR is the most-optimal state such that alternative ASRs can be rejected. The criterion for determining which genes belong in these categories was the parsimony optimization method implemented in Mesquite (v3.02). We thus identified 149 genes that unambiguously evolved endometrial expression in the stem-lineage of Eutherian mammals (Figure 1—source data 3).
Pathway enrichments
Request a detailed protocolWe used WebGestalt v. 2019 (Liao et al., 2019) to determine if the 149 identifies genes were enriched in ontology terms using over-representation analysis (ORA). A key advantage of WebGestalt is that it allowed for the inclusion of a custom background gene list, which was the set of 21,750 genes for which we could reconstruct ancestral states, rather than all annotated protein-coding genes in the human genome. We used ORA to identify enriched terms for three pathway databases (KEGG, Reactome, and Wikipathway), the Human Phenotype Ontology database, and a custom database of genes implicated in preterm birth by GWAS. The preterm birth gene set was assembled from the NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog), including genes implicated in GWAS with either the ontology terms ‘Preterm Birth’ (EFO_0003917) or ‘Spontaneous Preterm Birth’ (EFO_0006917), as well as two recent preterm birth GWAS (Warrington et al., 2019; Sakabe et al., 2020) using a genome-wide significant p-value of 9 × 10−6. The custom gmt file used to test for enrichment of preterm birth associated genes is included as a supplementary data file to Figure 1 (Figure 1—source data 8).
Marsupial gene expression analysis
Opossum uterine gene expression time course
Request a detailed protocolTo explore the expression of HAND2 and other genes throughout gestation in Marsupials, we analyzed previously generated short-tailed opossum (Monodelphis domestica) RNA-Seq data from virgin non-pregnant (SRR2972837, SRR2972848), day 7 (during the histotrophic phase; SRP111668), day 12.5 (just after hatching and during the transition from the histotrophic to the placental phase; GSM1611397), day 13 (early placental phase; SRP111668), day 13.5–14.5 (during the late placental to early parturition phase; SRR2969483, SRR2969536, SRR2970443), and 9–10 month post-partum utera (SRR2972728 and SRR2972729) (Figure 1—source data 7; Lynch et al., 2015; Hansen et al., 2016; Griffith et al., 2017; Griffith et al., 2019).
Wallaby RNA-Seq data from non-pregnant and pregnant animals
Request a detailed protocolRNA-Seq data for tammar wallaby (Macropus eugenii) uterine tissue from pregnant and non-pregnant animals were from PRJDB1934.
RNA-Seq analysis
Request a detailed protocolWe used Kallisto version 0.42.4 to pseudo-align RNA-Seq reads to the M. domestica and M. eugenii reference transcriptomes with default parameters bias correction and 100 bootstrap replicates. Kallisto output quantifies transcript abundance estimates in TPM.
Immunohistochemistry (IHC)
Request a detailed protocolEndometrial tissue from pregnant opossum (12.5d) was fixed in 10% neutral-buffered formalin, paraffin-embedded, sectioned at 4 μm, and mounted on slides. Paraffin sections were dried at room temperature overnight and then baked for 12 hr at 50°C. Prior to immunostaining, de-paraffinization and hydration were done in xylene and graded ethanol to distilled water. During hydration, a 5 min blocking for endogenous peroxidase was done in 0.3% H2O2 in 95% ethanol. Antigen retrieval was performed in retrieval buffer pH 6, using a pressure boiler microwave as a heat source with power set to full, allowing retrieval buffer to boil for 20 min, and then cooled in a cold water bath for 10 min. To stain sections, we used the Pierce Peroxidase IHC Detection Kit (cat # 36000) following the manufacturer's protocol. Briefly, uterine sections were incubated at 4°C overnight with polyclonal antibodies against HAND2 (Santa Cruz SC-9409), MUC1 (Novus Biologicals NB120-15481), p-ERα (Santa Cruz SC-12915), p-Erk1/2 (also known as MAPK1/2; Santa Cruz SC-23759-R) at 1:1000 dilution in blocking buffer. The next day sections were washed 3x in wash buffer, and incubated with HRP-conjugated rabbit anti-mouse IgG (H+L) secondary antibody (Invitrogen cat # 31450) at 1:10,000 dilution in blocking buffer. After 30 min at 4°C, slides were washed 3x in wash buffer. Slides were developed with 1x DAB/metal concentrate and stable peroxide buffer for 5 min, then rinsed 3x for 3 min in wash buffer, and mounted with Permount (SP15-100; Thermo Fisher Scientific).
Expression of HAND2 and IL15 at the maternal-fetal interface
Request a detailed protocolWe used previously published single-cell RNA-Seq (scRNA-Seq) data from the human first trimester maternal-fetal interface (Vento-Tormo et al., 2018) to determine which cell types express HAND2 and IL15. The dataset consists of transcriptomes for ~70,000,000 individual cells of many different cell types, including: three populations of tissue resident decidual natural killer cells (dNK1, dNK2, and dNK3), a population of proliferating natural killer cells (dNKp), type two and/or type three innate lymphoid cells (ILC2/ILC3), three populations of decidual macrophages (dM1, dM2, and dM3), two populations of dendritic cells (DC1 and DC2), granulocytes (Gran), T cells (TCells), maternal and lymphatic endothelial cells (Endo), two populations of epithelial glandular cells (Epi1 and Epi2), two populations of perivascular cells (PV1 and PV2), two endometrial stromal fibroblast populations (ESF1 and ESF2), and decidual stromal cells (DSCs), placental fibroblasts (fFB1), extravillous- (EVT), syncytio- (SCT), and villus- (VCT) cytotrophoblasts (Figure 2A). Data were not reanalyzed, rather previously analyzed data were accessed using the cell×gene website available at https://maternal-fetal-interface.cellgeni.sanger.ac.uk.
We note that Vento-Tormo et al. identified five populations of cells in the endometrial stromal lineage, including two perivascular populations (likely reflecting the mesenchymal stem cell-like progenitor of endometrial stromal fibroblasts and decidual stromal cells) and three cell types they call ‘decidual stromal cells’ and label ‘dS1-3’. However, based on the gene expression patterns of ‘dS1-3’ (shown in Vento-Tormo et al. Figure 3a), only ‘dS3’ are decidualized, as indicated by expression of classical markers of decidualization such and PRL (Tabanelli et al., 1992) and IGFBP1/2/6 (Tabanelli et al., 1992; Kim et al., 1999). In stark contrast, ‘dS1’ do not express decidualization markers but highly express markers of ESFs such as TAGLN and ID2, as well as markers of proliferating ESFs including ACTA2 (Kim et al., 1999). ‘dS2’ also express ESFs markers (TAGLN, ID2, ACTA2), but additionally LEFTY2 and IGFBP1/2/6, consistent with ESFs that have initiated the process of decidualization. These data indicate that the ‘dS1’ and ‘dS2’ populations are both ESFs, but ‘dS2’ are ESFs that have initiated decidualization (because they express IGFBPs but not PRL), and that ‘dS3’ are DSCs. Vento-Tormo et al. show that the differences in gene expression between ‘dS1-3’ are related to their topography in the endometrium, but degree of decidualization (‘dS1’/ESF1 < ‘dS2’/ESF2 < ‘dS3’/DSC) is also linked to differential gene expression.
Consistent with this, other scRNA-Seq studies have identified two ESF populations and one DSC population in the first trimester decidua, and used pseudotime analyses to show that they represent different states of differentiation from ESFs to mature DSCs (Suryawanshi et al., 2018). Therefore, we prefer to use the ESF1/ESF2/DSC nomenclature because it more accurately reflects the biology and gene expression profile of these cell types than the ‘dS1-3’ naming convention. We also note that while it is generally thought that ESFs are absent from the pregnant uterus, ESFs retain a presence in the endometrium from the first trimester until term (Richards et al., 1995; Suryawanshi et al., 2018; Muñoz-Fernández et al., 2019; Sakabe et al., 2020).
Expression of HAND2 and IL15 in human decidual cells
Request a detailed protocolWe used previously published IHC data for HAND2 and IL15 generated from pregnant human decidua as part of the Human Protein Atlas project (http://www.proteinatlas.org/; Uhlén et al., 2015). Image/gene/data available from IL15 (https://www.proteinatlas.org/ENSG00000164136-IL15/tissue) and HAND2 (https://www.proteinatlas.org/ENSG00000164107-HAND2/tissue).
Functional genomic analyses of the HAND2 and IL15 loci
Gene expression data
Request a detailed protocolWe used previously published RNA-Seq and microarray gene expression data generated from human ESFs and DSCs that were downloaded from National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA) and processed remotely using Galaxy platform (https://usegalaxy.org/; Version 20.01) (Afgan et al., 2018) for RNA-Seq data and GEO2R. RNA-Seq datasets were transferred from SRA to Galaxy using the Download and Extract Reads in FASTA/Q format from NCBI SRA tool (version 2.10.4+galaxy1). We used HISAT2 (version 2.1.0+galaxy5; Kim et al., 2015) to align reads to the Human hg38 reference genome using single- or paired-end options depending on the dataset and unstranded reads, and report alignments tailored for transcript assemblers including StringTie. Transcripts were assembled and quantified using StringTie (v1.3.6) (Pertea et al., 2015; Pertea et al., 2016), with reference file to guide assembly and the ‘reference transcripts only’ option, and output count files for differential expression with DESeq2/edgeR/limma-voom. Differentially expressed genes were identified using DESeq2 (version 2.11.40.6+galaxy1) (Anders and Huber, 2010; Love et al., 2014). The reference file for StringTie guided assembly was wgEncodeGencodeBasicV33. GEO2R performs comparisons on original submitter-supplied processed data tables using the GEOquery (Davis and Meltzer, 2007) and limma (Smyth et al., 2002) R packages from the Bioconductor project (https://bioconductor.org/; Gentleman et al., 2004).
Datasets included gene expression profiles of primary human ESFs treated for 48 hr with control non-targeting, PGR-targeting (GSE94036), FOXO1-targeting (GSE94036) or NR2F2 (COUP-TFII)-targeting (GSE47052) siRNA prior to decidualization stimulus for 72 hr; transfection with GATA2-targeting siRNA was followed immediately by decidualization stimulus (GSE108407). We also explored the expression of HAND2 and IL5 in the endometria of women with recurrent spontaneous abortion and implantation failure using a previously published dataset (GSE26787), as well as in the endometrium throughout the menstrual cycle (GSE4888) and basal plate throughout gestation (GSE5999); HAND2 probe 220480_at, IL15 probe 205992_s_at. The expression of HAND2 and IL5 in ESFs and DSCs from women with normal pregnancy and severe preeclampsia was assessed from a previously generated Agilent Whole Human Genome Microarray 4 × 44K v2 dataset (GSE91077).
ChIP-Seq and open chromatin data
Request a detailed protocolWe used previously published ChIP-Seq data generated from human DSCs that were downloaded from NCBI SRA and processed remotely using Galaxy (Afgan et al., 2018). ChIP-Seq reads were mapped to the human genome (GRCh37/hg19) using HISAT2 (Kim et al., 2015) with default parameters and peaks called with MACS2 (Zhang et al., 2008; Feng et al., 2012) with default parameters. Samples included PLZF (GSE75115), H3K4me3 (GSE61793), H3K27ac (GSE61793), H3K4me1 (GSE57007), PGR (GSE69539), the PGR A and B isoforms (GSE62475), NR2F2 (GSE52008), FOSL2 (GSE69539), FOXO1 (GSE69542), PolII (GSE69542), GATA2 (GSE108408), SRC-2/NCOA2 (GSE123246), AHR (GSE118413), ATAC-Seq (GSE104720), and DNase1-Seq (GSE61793). FAIRE-Seq peaks were downloaded from the UCSC genome browser and not re-called.
Chromatin interaction data
Request a detailed protocolTo assess chromatin looping, we utilized a previously published H3K27ac HiChIP dataset from a normal hTERT-immortalized endometrial cell line (E6E7hTERT) and three endometrial cancer cell lines (ARK1, Ishikawa and JHUEM-14) (O’Mara et al., 2019). This study identified 66,092 to 449,157 cis HiChIP loops (5 kb–2 Mb in length) per cell line. The majority of loops involved interactions of over 20 kb in distance, 35–40% of loops had contact with a promoter and those promoter-associated loops had a median span >200 kb. Contact data were from the original publication and not re-called for this study. Note that Figures 2C and 4C were made using a combination of the UCSC genome browser to map the location of regions of open chromatin and ChIP-Seq peaks and Illustrator to simplify the images from the genome browser.
Cell culture and HAND2 knockdown
Request a detailed protocolHuman hTERT-immortalized endometrial stromal fibroblasts (T-HESC; CRL-4003, ATCC) were grown in maintenance medium, consisting of Phenol Red-free DMEM (31053–028, Thermo Fisher Scientific), supplemented with 10% charcoal-stripped fetal bovine serum (CS-FBS; 12676029, Thermo Fisher Scientific), 1% L-glutamine (25030–081, Thermo Fisher Scientific) , 1% sodium pyruvate (11360070, Thermo Fisher Scientific), and 1x insulin-transferrin-selenium (ITS; 41400045, Thermo Fisher Scientific). A total of 2 × 105 cells were plated per well of a six-well plate and 18 hr later cells in 1750 μl of Opti-MEM (31985070, Thermo Fisher Scientific) were transfected with 50 nM of siRNA targeting HAND2 (s18133; Silencer Select Pre-Designed siRNA; cat # 4392420, Thermo Fisher Scientific) and 9 μl of Lipofectamine RNAiMAX (133778–150, Invitrogen) in 250 μl Opti-MEM. BlockIT Fluorescent Oligo (44–2926, Thermo Fisher Scientific) was used as a scrambled non-targeting RNA control. Cells were incubated in the transfection mixture for 6 hr. Then, cells were washed with PBS and incubated in the maintenance medium overnight. Cells in the control wells were checked under the microscope for fluorescence the next day. Forty-eight hr post-treatment, cells were washed with PBS, trypsinized (0.05% Trypsin-EDTA; 15400–054, Thermo Fisher Scientific) and total RNA was extracted using RNeasy Plus Mini Kit (74134, QIAGEN) following the manufacturer’s protocol. The knockdown experiment was done in three biological replicates. To test for the efficiency of the knockdown, cDNA was synthesized from 100 to 200 ng RNA using Maxima H Minus First Strand cDNA Synthesis Kit (K1652, Thermo Fisher Scientific) following the manufacturer’s protocol. qRT-PCR was performed using QuantiTect SYBR Green PCR (204143, QIAGEN). HAND2 primers: forward CACCAGCTACATCGCCTACC, reverse ATTTCGTTCAGCTCCTTCTTCC. GAPDH housekeeping gene was used for normalization; primers forward AATCCCATCACCATCTTCCA, reverse TGGACTCCACGACGTACTCA. Samples that showed >70% knockdown efficiency were used for RNA-Seq.
HAND2 knockdown RNA-Seq analysis
Request a detailed protocolRNA from knockdown and control samples were DNase treated with TURBO DNA-free Kit (AM1907, Thermo Fisher Scientific) and RNA quality and quantity were assessed on 2100 Bioanalyzer (Agilent Technologies, Inc). RNA-Seq libraries were prepared using TruSeq Stranded Total RNA Library Prep Kit with Ribo-Zero Human (RS-122–2201, Illumina Inc) following manufacturer’s protocol. Library quality and quantity were checked on 2100 Bioanalyzer and the pool of libraries was sequenced on Illumina HiSEQ4000 (single-end 50 bp) using manufacturer’s reagents and protocols. Quality control, Ribo-Zero library preparation and Illumina sequencing were performed at the Genomics Facility at The University of Chicago.
All sequencing data were uploaded and analyzed on the Galaxy platform (https://usegalaxy.org/; Version 20.01). Individual reads for particular samples were concatenated using the ‘Concatenate datasets’ tool (version 1.0.0). We used HISAT2 (version 2.1.0+galaxy5) (Kim et al., 2015) to align reads to the Human hg38 reference genome using ‘Single-end’ option, and reporting alignments tailored for transcript assemblers including StringTie. Transcripts were assembled and quantified using StringTie (v1.3.6) (Pertea et al., 2015; Pertea et al., 2016), with reference file to guide assembly and the ‘reference transcripts only’ option, and output count files for differential expression with DESeq2/edgeR/limma-voom. Differentially expressed genes were identified using DESeq2 (version 2.11.40.6+galaxy1; Anders and Huber, 2010; Love et al., 2014). The reference file for StringTie guided assembly was wgEncodeGencodeBasicV33. The volcano plot was generated using Blighe et al., 2020.
Trans-well migration assay
Request a detailed protocolHuman hTERT-immortalized ESFs (CRL-4003, ATCC) were selected as a model ESF cell line because they are proliferative, maintain hormone responsiveness, and gene expression patterns characteristic of primary ESFs, and have been relatively well characterized (Krikun et al., 2004). ESFs were cultured in the maintenance medium as described above in T75 flasks until ~80% confluent. Cryopreserved primary adult human CD56+ NK cells purified by immunomagnetic bead separation were obtained from ATCC (PCS-800–019) and cultured in RPMI-1640 containing 10% FBS and 500 IU/ml IL2 in T75 flasks for 2 days prior to trans-well migration assays. We used the immortalized first trimester extravillous trophoblast cell line HTR-8/SVneo (Graham et al., 1993), because it maintains characteristics of extravillous trophoblasts and has previously been shown to be a good model of trophoblast migration and invasion (Iacob et al., 2008; Paiva et al., 2009; Hannan et al., 2010). HTR-8/SVneo cells were obtained from ATCC (CRL-3271) and cultured in RPMI-1640 containing 5% FBS in T75 flasks for 2 days prior to trans-well migration assays. All cell lines were determined to be mycoplasma free before each experiment.
A total of 3 × 104 ESFs were plated per well of a 24-well plate and 18 hr later cells were transfected in Opti-MEM with 10 nM (per well) of siRNA targeting HAND2 (s18133; Silencer Select Pre-Designed siRNA, cat # 4392420; Thermo Fisher Scientific) or IL15 (s7377; Silencer Select Pre-Designed siRNA, cat # 4392420; Thermo Fisher Scientific) and 1.5 μl (per well) of Lipofectamine RNAiMAX (133778–150; Invitrogen). As a negative control we used Silencer Select negative control No. 1 (4390843; Thermo Fisher Scientific). ESFs were incubated in the transfection mixture for 6 hr. Then, ESFs were washed with warm PBS and incubated in the maintenance medium overnight. Efficiency of the knockdown was confirmed 48 hr post-treatment by qRT-PCR, media from each well was transferred to new 24-well plates and stored at 4°C. Total RNA from cells was extracted using RNeasy Plus Mini Kit (74134, QIAGEN) following the manufacturer’s protocol. cDNA was synthesized from 10 ng RNA using Maxima H Minus First Strand cDNA Synthesis Kit (K1652, Thermo Fisher Scientific) following the manufacturer’s protocol. qRT-PCR was performed using TaqMan Fast Universal PCR Master Mix 2X (4352042, Thermo Fisher Scientific), with primers for HAND2 (Hs00232769_m1), IL15 (Hs01003716_m1), and Malat (Hs00273907_s1) as a control housekeeping gene. Conditioned media from samples with >70% knockdown efficiency was used for trans-well migration assays.
Corning HTS Trans-well permeable supports were used for the trans-well migration assay (Corning, cat # CLS3398). Prior to the assays, 500 μl of media conditioned for 12 hr was centrifuged for 3 min at 1000 RPM to pellet any cells. For experiments using recombinant human IL15 (rhIL15), we added 10 ng/ml (AbCam, ab259403) of rhIL15 to fresh, non-conditioned ESF media; 10 ng/ml has previously been shown to induce migration of JEG-3 choriocarcinoma cells (Zygmunt et al., 1998). For neutralizing antibody experiments, either 1 μg/ml of anti-IL15 IgG (AbCam cat # MA5-23729) or control IgG (AbCam cat # 31903) were added to non-conditioned ESF media; 1 μg/ml has previously been shown to neutralize ESF-derived proteins and inhibit AC-1M88 trophoblast cell migration in trans-well assays (Gellersen et al., 2010; Gellersen et al., 2013). Plates were incubated with shaking at 37°C for 30 min prior to initiation of migration assays. During antibody incubation, NK and HTR-8/SVneo cells were collected and resuspended in fresh ESF growth media. For the trans-well migration assay, 5 × 106 of either NK or HTR-8/SVneo cells were added to each well of the upper chamber and either treatment or control media were added to the lower chambers. Plates were incubated at 37°C with 5% CO2.
After 8 hr incubation, we removed the upper plate (containing remaining NK and HTR-8/SVneo cells) and discarded non-migrated cells. Fifty μl from each well in the lower chamber was transferred into a single well of a 96-well opaque plate. We used the CellTiter-Glo luminescent cell viability assay (G7570, Promega) to measure luminescence, which is proportional to the number of live cells per well. Data are reported as effect sizes (mean differences) between treatment and control. Confidence intervals are bias-corrected and accelerated. The p-values reported are the likelihoods of the observed effect sizes, if the null hypothesis of zero difference is true and calculated from a two-sided permutation t-test (5000 reshuffles of the control and test labels). Cumming estimation plots and estimation statistics were calculated using DABEST R package (Ho et al., 2019).
Data availability
Sequencing data have been deposited in GEO under accession codes GSE155170 and GSE155322.
-
NCBI Gene Expression OmnibusID GSM5100045. Mouse Endometrium Individual 1.
-
NCBI Gene Expression OmnibusID GSM5100046. Mouse Endometrium Individual 2.
-
NCBI Gene Expression OmnibusID GSM5100047. Mouse Endometrium Individual 3.
-
NCBI Gene Expression OmnibusID GSM4696515. Baboon Endometrium Individual 1.
-
NCBI Gene Expression OmnibusID GGSM4696516. Baboon Endometrium Individual 2.
-
NCBI Gene Expression OmnibusID GSM4696517. Baboon Endometrium Individual 3.
-
NCBI Gene Expression OmnibusID GSM4696518. Hamster Endometrium Individual 1 Replicate 1.
-
NCBI Gene Expression OmnibusID GSM4696519. Hamster Endometrium Individual 2 Replicate 1.
-
NCBI Gene Expression OmnibusID GSM4696520. Hamster Endometrium Individual 3 Replicate 1.
-
NCBI Gene Expression OmnibusID GSM4696521. Hamster Endometrium Individual 1 Replicate 2.
-
NCBI Gene Expression OmnibusID GSM4696522. Hamster Endometrium Individual 2 Replicate 2.
-
NCBI Gene Expression OmnibusID GSM4696523. Hamster Endometrium Individual 3 Replicate 2.
-
NCBI Gene Expression OmnibusID GSM4696524. Bat Endometrium Individual 1.
-
NCBI Gene Expression OmnibusID GSM4696525. Bat Endometrium Individual 2.
-
NCBI Gene Expression OmnibusID GSM4696526. Squirrel Endometrium Individual 1.
-
NCBI Gene Expression OmnibusID GSM4696527. Squirrel Endometrium Individual 2.
-
NCBI Gene Expression OmnibusID GSM4699113. siRNA Ctrl_rep1a.
-
NCBI Gene Expression OmnibusID GSM4699114. siRNA Ctrl_rep1b.
-
NCBI Gene Expression OmnibusID GSM4699115. siRNA Ctrl_rep2a.
-
NCBI Gene Expression OmnibusID GSM4699116. siRNA Ctrl_rep2b.
-
NCBI Gene Expression OmnibusID GSM4699117. siRNA Ctrl_rep3a.
-
NCBI Gene Expression OmnibusID GSM4699118. siRNA Ctrl_rep3b.
-
NCBI Gene Expression OmnibusID GSM4699119. siRNA HAND2_rep1a.
-
NCBI Gene Expression OmnibusID GSM4699120. siRNA HAND2_rep1b.
-
NCBI Gene Expression OmnibusID GSM4699121. siRNA HAND2_rep2a.
-
NCBI Gene Expression OmnibusID GSM4699122. siRNA HAND2_rep2b.
-
NCBI Gene Expression OmnibusID GSM4699123. siRNA HAND2_rep3a.
-
NCBI Gene Expression OmnibusID GSM4699124. siRNA HAND2_rep3b.
-
NCBI Gene Expression OmnibusID GSE155170. Evolutionary transcriptomics implicates HAND2 in the origins of implantation and regulation of gestation length.
-
NCBI Gene Expression OmnibusID GSE155322. Evolutionary transcriptomics implicates HAND2 in the origins of implantation and regulation of gestation length.
References
-
Adverse reproductive outcomes in the transgenic Ah receptor-deficient mouseToxicology and Applied Pharmacology 155:62–70.https://doi.org/10.1006/taap.1998.8601
-
The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 updateNucleic Acids Research 46:W537–W544.https://doi.org/10.1093/nar/gky379
-
IL-15 is chemotactic for natural killer cells and stimulates their adhesion to vascular endotheliumJournal of Leukocyte Biology 61:729–735.https://doi.org/10.1002/jlb.61.6.729
-
Molecular evolution of genes associated with preeclampsia: genetic conflict, antagonistic coevolution and signals of selection’Journal of Evolutionary Medicine 6:msq034.https://doi.org/10.1093/molbev/msq034
-
Quantification of the predominant immune cell populations in decidua throughout human pregnancyAmerican Journal of Reproductive Immunology 71:109–119.https://doi.org/10.1111/aji.12185
-
Near-optimal probabilistic RNA-seq quantificationNature Biotechnology 34:525–527.https://doi.org/10.1038/nbt.3519
-
Uterine NK cells, spiral artery modification and the regulation of blood pressure during mouse pregnancyAmerican Journal of Reproductive Immunology 63:472–481.https://doi.org/10.1111/j.1600-0897.2010.00818.x
-
FOXO1A differentially regulates genes of decidualizationEndocrinology 147:3870–3876.https://doi.org/10.1210/en.2006-0167
-
Effect of human endometrial stromal cell-derived conditioned medium on uterine natural killer (uNK) cells' proliferation and cytotoxicityAmerican Journal of Reproductive Immunology 65:589–596.https://doi.org/10.1111/j.1600-0897.2010.00955.x
-
IL-15 regulation in human endometrial stromal cellsThe Journal of Clinical Endocrinology & Metabolism 87:1898–1901.https://doi.org/10.1210/jcem.87.4.8539
-
Evolutionary origins of preeclampsiaPregnancy Hypertension: An International Journal of Women's Cardiovascular Health 7:56.https://doi.org/10.1016/j.preghy.2016.10.006
-
Mechanisms of T cell tolerance towards the allogeneic fetusNature Reviews Immunology 13:23–33.https://doi.org/10.1038/nri3361
-
Uterine natural killer cell partnerships in early mouse decidua basalisJournal of Leukocyte Biology 100:645–655.https://doi.org/10.1189/jlb.1HI0515-226R
-
Identifying ChIP-seq enrichment using MACSNature Protocols 7:1728–1740.https://doi.org/10.1038/nprot.2012.101
-
Decidual natural killer cells regulate vessel stability: implications for impaired spiral artery remodellingJournal of Reproductive Immunology 110:54–60.https://doi.org/10.1016/j.jri.2015.04.003
-
HAND2-mediated proteolysis negatively regulates the function of estrogen receptor αMolecular Medicine Reports 12:5538–5544.https://doi.org/10.3892/mmr.2015.4070
-
Inhibition of allogeneic T-cell responses by dendritic cells expressing transduced indoleamine 2,3-dioxygenaseThe Journal of Gene Medicine 7:565–575.https://doi.org/10.1002/jgm.698
-
Cyclic AMP and progesterone receptor cross-talk in human endometrium: a decidualizing affairJournal of Endocrinology 178:357–372.https://doi.org/10.1677/joe.0.1780357
-
The reptilian oviduct: a review of structure and function and directions for future researchJournal of Experimental Zoology 293:141–170.https://doi.org/10.1002/jez.10105
-
Decidualized endometrial stromal cell derived factors promote trophoblast invasionFertility and Sterility 95:1278–1283.https://doi.org/10.1016/j.fertnstert.2010.09.045
-
Regulation of decidualization, interleukin-11 and interleukin-15 by homeobox A 10 in endometrial stromal cellsJournal of Reproductive Immunology 85:130–139.https://doi.org/10.1016/j.jri.2010.03.003
-
Uterine natural killer cells dysregulation in idiopathic human preterm birth: a pilot studyThe Journal of Maternal-Fetal & Neonatal Medicine 30:1782–1786.https://doi.org/10.1080/14767058.2016.1224840
-
Invasion of the leukocytes into the fetal-maternal interface during pregnancyJournal of Leukocyte Biology 88:625–633.https://doi.org/10.1189/jlb.1209796
-
Establishment and characterization of first trimester human trophoblast cells with extended lifespanExperimental Cell Research 206:204–211.https://doi.org/10.1006/excr.1993.1139
-
Mechanism of control of trophoblast invasion in situJournal of Cellular Physiology 148:228–234.https://doi.org/10.1002/jcp.1041480207
-
Reproductive endocrinology: circadian clock involved in embryo implantationNature Reviews. Endocrinology 10:701.https://doi.org/10.1038/nrendo.2014.174
-
Endometrial recognition of pregnancy occurs in the grey short-tailed opossum (Monodelphis domestica)Proceedings of the Royal Society B: Biological Sciences 286:20190691.https://doi.org/10.1098/rspb.2019.0691
-
AGEs, contributors to placental bed vascular changes leading to preeclampsiaFree Radical Research 47:70–80.https://doi.org/10.3109/10715762.2013.815347
-
Models for study of human embryo implantation: choice of cell lines?Biology of Reproduction 82:235–245.https://doi.org/10.1095/biolreprod.109.077800
-
Possible role of the 'IDO-AhR axis' in maternal-foetal toleranceCell Biology International 37:105–108.https://doi.org/10.1002/cbin.10023
-
Wnt genes in the mouse uterus: potential regulation of implantationBiology of Reproduction 80:989–1000.https://doi.org/10.1095/biolreprod.108.075416
-
Vascular-leukocyte interactions: mechanisms of human decidual spiral artery remodeling in vitroAmerican Journal of Pathology 177:1017–1030.https://doi.org/10.2353/ajpath.2010.091105
-
Maternal activating KIRs protect against human reproductive failure mediated by fetal HLA-C2Journal of Clinical Investigation 120:4102–4110.https://doi.org/10.1172/JCI43998
-
The development of the Monotremata-Part I The histology of the oviduct during gestation by Catherine J Hill BSc, PhD part II the structure of the Egg-shellThe Transactions of the Zoological Society of London 21:413–476.https://doi.org/10.1111/j.1096-3642.1936.tb00458.x
-
Elephant transcriptome provides insights into the evolution of eutherian placentationGenome Biology and Evolution 4:713–725.https://doi.org/10.1093/gbe/evs045
-
The role of FOXO1 in the decidual transformation of the endometrium and early pregnancyMedical Molecular Morphology 46:61–68.https://doi.org/10.1007/s00795-013-0018-z
-
Blastocyst invasion and the stromal response in primatesHuman Reproduction 14:45–55.https://doi.org/10.1093/humrep/14.suppl_2.45
-
HISAT: a fast spliced aligner with low memory requirementsNature Methods 12:357–360.https://doi.org/10.1038/nmeth.3317
-
IL-15 expression at human endometrium and deciduaBiology of Reproduction 63:683–687.https://doi.org/10.1095/biolreprod63.3.683
-
Central role of interleukin-15 in postovulatory recruitment of peripheral blood CD16(-) natural killer cells into human endometriumThe Journal of Clinical Endocrinology & Metabolism 90:2932–2940.https://doi.org/10.1210/jc.2004-2447
-
Physiological role of IL-15 and IL-18 at the maternal-fetal interfaceChemical Immunology and Allergy 89:10–25.https://doi.org/10.1159/000087906
-
Bmp2 is critical for the murine uterine decidual responseMolecular and Cellular Biology 27:5468–5478.https://doi.org/10.1128/MCB.00342-07
-
Bone morphogenetic protein 2 functions via a conserved signaling pathway involving Wnt4 to regulate uterine decidualization in the mouse and the humanJournal of Biological Chemistry 282:31725–31732.https://doi.org/10.1074/jbc.M704723200
-
Transforming growth factor β signaling in uterine development and functionJournal of Animal Science and Biotechnology 5:52.https://doi.org/10.1186/2049-1891-5-52
-
WebGestalt 2019: gene set analysis toolkit with revamped UIs and APIsNucleic Acids Research 47:W199–W205.https://doi.org/10.1093/nar/gkz401
-
Leukocyte driven-decidual angiogenesis in early pregnancyCellular & Molecular Immunology 11:522–537.https://doi.org/10.1038/cmi.2014.63
-
The role of nuclear factor kappa B in human labourReproduction 130:569–581.https://doi.org/10.1530/rep.1.00197
-
A novel HAND2 loss-of-function mutation responsible for tetralogy of fallotInternational Journal of Molecular Medicine 37:445–451.https://doi.org/10.3892/ijmm.2015.2436
-
SoftwareMesquite: A Modular System for Evolutionary AnalysisMesquite: A Modular System for Evolutionary Analysis.
-
Estrogen induction of ovalbumin mRNA: evidence for transcription controlMolecular and Cellular Biochemistry 7:33–42.https://doi.org/10.1007/BF01732161
-
Novel concepts on pregnancy clocks and alarms: redundancy and synergy in human parturitionHuman Reproduction Update 22:535–560.https://doi.org/10.1093/humupd/dmw022
-
Progesterone withdrawal and estrogen activation in human parturition are coordinated by progesterone receptor A expression in the myometriumThe Journal of Clinical Endocrinology & Metabolism 87:2924–2930.https://doi.org/10.1210/jcem.87.6.8609
-
Natural killer cells and pregnancyNature Reviews Immunology 2:656–663.https://doi.org/10.1038/nri886
-
The transcription factor HAND2 up-regulates transcription of the IL15 gene in human endometrial stromal cellsJournal of Biological Chemistry 295:9596–9605.https://doi.org/10.1074/jbc.RA120.012753
-
Uterine NK cells mediate inflammation-induced fetal demise in IL-10-null miceThe Journal of Immunology 175:4084–4090.https://doi.org/10.4049/jimmunol.175.6.4084
-
Evidence for participation of uterine natural killer cells in the mechanisms responsible for spontaneous preterm labor and deliveryAmerican Journal of Obstetrics and Gynecology 200:308.e1–30308.https://doi.org/10.1016/j.ajog.2008.10.043
-
Tgfβ superfamily signaling and uterine decidualizationReproductive Biology and Endocrinology 15:0303.https://doi.org/10.1186/s12958-017-0303-0
-
Molecular regulation of parturition: the role of the decidual clockCold Spring Harbor Perspectives in Medicine 5:a023143.https://doi.org/10.1101/cshperspect.a023143
-
Expression of interleukin-15 in human endometrium and deciduaMolecular Human Reproduction 6:75–80.https://doi.org/10.1093/molehr/6.1.75
-
Interleukin-1 inhibits interleukin-15 production by progesterone during in vitro decidualization in humanJournal of Reproductive Immunology 61:3–12.https://doi.org/10.1016/j.jri.2003.10.002
-
Circadian aspects of mammalian parturition: a reviewMolecular and Cellular Endocrinology 349:62–67.https://doi.org/10.1016/j.mce.2011.06.041
-
Melatonin and the circadian timing of human parturitionReproductive Sciences 20:168–174.https://doi.org/10.1177/1933719112442244
-
Activation of the receptor for advanced glycation end products system in women with severe preeclampsiaThe Journal of Clinical Endocrinology & Metabolism 96:689–698.https://doi.org/10.1210/jc.2010-1418
-
StringTie enables improved reconstruction of a transcriptome from RNA-seq readsNature Biotechnology 33:290–295.https://doi.org/10.1038/nbt.3122
-
Influence of estradiol-17-beta upon the oxytocic action of oxytocin in the pregnant human uterusAmerican Journal of Obstetrics and Gynecology 96:857–862.https://doi.org/10.1016/0002-9378(66)90682-X
-
Preventing preterm birth: the past limitations and new potential of animal modelsDisease Models & Mechanisms 3:407–414.https://doi.org/10.1242/dmm.001701
-
Natural killer-cell deficiency alters placental development in ratsBiology of Reproduction 96:145–158.https://doi.org/10.1095/biolreprod.116.142752
-
BookReproduction in Monotremes and MarsupialsIn: Renfree M, editors. Encyclopedia of Life Sciences. Chichester, UK: John Wiley & Sons, Ltd. pp. 1–10.https://doi.org/10.1038/npg.els.0001856
-
Anti-inflammatory mediators as physiological and pharmacological regulators of parturitionExpert Review of Clinical Immunology 7:675–696.https://doi.org/10.1586/eci.11.58
-
Immune cells and preterm labour: do invariant NKT cells hold the key?Molecular Human Reproduction 21:309–312.https://doi.org/10.1093/molehr/gav002
-
Gland formation from human endometrial epithelial cells in vitroIn Vitro Cellular & Developmental Biology 24:1037–1041.https://doi.org/10.1007/BF02620878
-
Uterine natural killer cells initiate spiral artery remodeling in human pregnancyThe FASEB Journal 26:4876–4885.https://doi.org/10.1096/fj.12-210310
-
Myometrial immune cells contribute to term parturition, preterm labour and post-partum involution in miceJournal of Cellular and Molecular Medicine 17:90–102.https://doi.org/10.1111/j.1582-4934.2012.01650.x
-
Patterns of plasma corticotropin-releasing hormone, progesterone, estradiol, and estriol change and the onset of human laborThe Journal of Clinical Endocrinology & Metabolism 94:2066–2074.https://doi.org/10.1210/jc.2008-2257
-
Evidence for immune cell involvement in decidual spiral arteriole remodeling in early human pregnancyThe American Journal of Pathology 174:1959–1971.https://doi.org/10.2353/ajpath.2009.080995
-
SoftwareLimma: Linear Models for Microarray and RNA-Seq Data User’s Guide, version bioconductorLimma: Linear Models for Microarray and RNA-Seq Data User’s Guide.
-
A HAND2 loss-of-function mutation causes familial ventricular septal defect and pulmonary stenosisG3 Genes|Genomes|Genetics 6:987–992.
-
A single-cell survey of the human first-trimester placenta and deciduaScience Advances 4:eaau4788.https://doi.org/10.1126/sciadv.aau4788
-
Genomics of preterm birthCold Spring Harbor Perspectives in Medicine 5:a023127.https://doi.org/10.1101/cshperspect.a023127
-
In vitro decidualization of human endometrial stromal cellsThe Journal of Steroid Biochemistry and Molecular Biology 42:337–344.https://doi.org/10.1016/0960-0760(92)90137-8
-
Overdosage of Hand2 causes limb and heart defects in the human chromosomal disorder partial trisomy distal 4qHuman Molecular Genetics 22:2471–2481.https://doi.org/10.1093/hmg/ddt099
-
Pre-eclampsia and maternal-fetal conflictEvolution, Medicine, and Public Health 2018:217–218.https://doi.org/10.1093/emph/eoy029
-
Nothing in medicine makes sense, except in the light of evolutionJournal of Molecular Medicine 90:481–494.https://doi.org/10.1007/s00109-012-0900-5
-
On the apparent rarity of epithelial cancers in captive chimpanzeesPhilosophical Transactions of the Royal Society B: Biological Sciences 370:20140225.https://doi.org/10.1098/rstb.2014.0225
-
A model based criterion for gene expression calls using RNA-seq dataTheory in Biosciences 132:159–164.https://doi.org/10.1007/s12064-013-0178-3
-
Decidual natural killer cell interactions with trophoblasts are impaired in pregnancies at increased risk of preeclampsiaThe American Journal of Pathology 183:1853–1861.https://doi.org/10.1016/j.ajpath.2013.08.023
-
Wnt6 is essential for stromal cell proliferation during decidualization in Mice1Biology of Reproduction 88:104687.https://doi.org/10.1095/biolreprod.112.104687
-
The progesterone receptor regulates implantation, decidualization, and glandular development via a complex paracrine signaling networkMolecular and Cellular Endocrinology 357:108–118.https://doi.org/10.1016/j.mce.2011.10.028
-
Decidual leucocyte populations in early to late gestation normal human pregnancyJournal of Reproductive Immunology 82:24–31.https://doi.org/10.1016/j.jri.2009.08.001
-
Progesterone signaling in myometrial cells: role in human pregnancy and parturitionCurrent Opinion in Physiology 13:117–122.https://doi.org/10.1016/j.cophys.2019.09.007
-
Oestrogen receptors in the genital tract of the Australian marsupial Trichosurus vulpeculaGeneral and Comparative Endocrinology 46:417–427.https://doi.org/10.1016/0016-6480(82)90095-8
-
Model-based analysis of ChIP-Seq (MACS)Genome Biology 9:R137.https://doi.org/10.1186/gb-2008-9-9-r137
-
Invasion of cytotrophoblastic (JEG-3) cells is up-regulated by interleukin-15 in vitroAmerican Journal of Reproductive Immunology 40:326–331.https://doi.org/10.1111/j.1600-0897.1998.tb00061.x
Article and author information
Author details
Funding
Burroughs Wellcome Fund (Preterm Birth Initiative 1013760)
- Vincent J Lynch
March of Dimes Foundation (UChicago-Northwestern-Duke Prematurity Research Center)
- Vincent J Lynch
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
The authors thank the following researchers for providing pregnant endometrial samples: GP Wagner (Yale University) – Monodelphis domestica; RR Behringer (The University of Texas MD Anderson Cancer Center) – Carollia perspicillata; BC Paria (Vanderbilt University School of Medicine) – Mesocricetus auratus, Mus musculus; AT Fazleabas (Michigan State University) – Papio anubis; DK Merriman (University of Wisconsin Oshkosh) – Ictidomys tridecemlineatus. We are also grateful to AM Bamberger (University Hospital Eppendorf) for providing the trans-well migration assay protocol, D Glubb (QIMR Berghofer Medical Research Institute) for assistance in interpreting the HiChIP assay data, R Beaumont (University of Exeter Medical School) and RM Freathy (University of Exeter) for assistance with interpreting maternal-fetal birth weight GWAS data, and VL Hansen (University of New Mexico) for providing details on stages of opossum RNA-Seq data. VJL thanks the Department of Human Genetics at The University of Chicago for support during the planning and preliminary data generation phase of this work. MM thanks Michael Sulak for the help with editing the manuscript.
Version history
- Received: July 20, 2020
- Accepted: January 29, 2021
- Accepted Manuscript published: February 1, 2021 (version 1)
- Version of Record published: March 9, 2021 (version 2)
Copyright
© 2021, Marinić et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 2,625
- views
-
- 384
- downloads
-
- 33
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Evolutionary Biology
- Genetics and Genomics
Comparing the genes expressed at the maternal-fetal interface in different species helps to pinpoint those that contribute to a healthy pregnancy by regulating the activity of the immune system.
-
- Evolutionary Biology
- Epidemiology and Global Health
- Microbiology and Infectious Disease
- Genetics and Genomics
eLife is pleased to present a Special Issue to highlight recent advances in the growing and increasingly interdisciplinary field of evolutionary medicine.






